Abstract
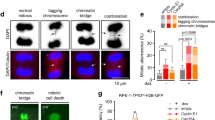
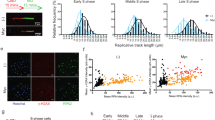
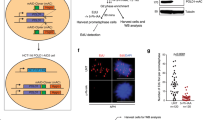
It has become increasingly clear that oncogenes not only provide aberrant growth signals to cells but also cause DNA damage at replication forks (replication stress), which activate the ataxia telangiectasia mutated (ATM)/p53-dependent tumor barrier. Here we studied underlying mechanisms of oncogene-induced replication stress in cells overexpressing the oncogene Cyclin E. Cyclin E overexpression is associated with increased firing of replication origins, impaired replication fork progression and DNA damage that activates RAD51-mediated recombination. By inhibiting replication initiation factors, we show that Cyclin E-induced replication slowing and DNA damage is a consequence of excessive origin firing. A significant amount of Cyclin E-induced replication slowing is due to interference between replication and transcription, which also underlies the activation of homologous recombination. Our data suggest that Cyclin E-induced replication stress is caused by deregulation of replication initiation and increased interference between replication and transcription, which results in impaired replication fork progression and DNA damage triggering the tumor barrier or cancer-promoting mutations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bartkova J, Rezaei N, Liontos M, Karakaidos P, Kletsas D, Issaeva N et al. Oncogene-induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 2006; 444: 633–637.
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K et al. DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 2005; 434: 864–870.
Di Micco R, Fumagalli M, Cicalese A, Piccinin S, Gasparini P, Luise C et al. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper-replication. Nature 2006; 444: 638–642.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 2005; 434: 907–913.
Halazonetis TD, Gorgoulis VG, Bartek J . An oncogene-induced DNA damage model for cancer development. Science 2008; 319: 1352–1355.
Negrini S, Gorgoulis VG, Halazonetis TD . Genomic instability—an evolving hallmark of cancer. Nat Rev Mol Cell Biol 2010; 11: 220–228.
Dominguez-Sola D, Ying CY, Grandori C, Ruggiero L, Chen B, Li M et al. Non-transcriptional control of DNA replication by c-Myc. Nature 2007; 448: 445–451.
Malumbres M, Barbacid M . To cycle or not to cycle: a critical decision in cancer. Nat Rev Cancer 2001; 1: 222–231.
Bester AC, Roniger M, Oren YS, Im MM, Sarni D, Chaoat M et al. Nucleotide deficiency promotes genomic instability in early stages of cancer development. Cell 2011; 145: 435–446.
Bartek J, Lukas J, Bartkova J . DNA damage response as an anti-cancer barrier: damage threshold and the concept of ‘conditional haploinsufficiency’. Cell Cycle 2007; 6: 2344–2347.
Mechali M . Eukaryotic DNA replication origins: many choices for appropriate answers. Nat Rev Mol Cell Biol 2010; 11: 728–738.
Jackson DA, Pombo A . Replicon clusters are stable units of chromosome structure: evidence that nuclear organization contributes to the efficient activation and propagation of S phase in human cells. J Cell Biol 1998; 140: 1285–1295.
Gilbert DM . Replication origin plasticity, Taylor-made: inhibition vs recruitment of origins under conditions of replication stress. Chromosoma 2007; 116: 341–347.
Woo RA, Poon RY . Cyclin-dependent kinases and S phase control in mammalian cells. Cell Cycle 2003; 2: 316–324.
Shechter D, Costanzo V, Gautier J . ATR and ATM regulate the timing of DNA replication origin firing. Nat Cell Biol 2004; 6: 648–655.
Sorensen CS, Syljuasen RG, Lukas J, Bartek J . ATR claspin and the Rad9-Rad1-Hus1 complex regulate Chk1 and Cdc25A in the absence of DNA damage. Cell Cycle 2004; 3: 941–945.
Syljuasen RG, Sorensen CS, Hansen LT, Fugger K, Lundin C, Johansson F et al. Inhibition of human Chk1 causes increased initiation of DNA replication, phosphorylation of ATR targets, and DNA breakage. Mol Cell Biol 2005; 25: 3553–3562.
Fang Y, Tsao CC, Goodman BK, Furumai R, Tirado CA, Abraham RT et al. ATR functions as a gene dosage-dependent tumor suppressor on a mismatch repair-deficient background. Embo J 2004; 23: 3164–3174.
Lam MH, Liu Q, Elledge SJ, Rosen JM . Chk1 is haploinsufficient for multiple functions critical to tumor suppression. Cancer Cell 2004; 6: 45–59.
Petermann E, Woodcock M, Helleday T . Chk1 promotes replication fork progression by controlling replication initiation. Proc Natl Acad Sci USA 2010; 107: 16090–16095.
Hwang HC, Clurman BE . Cyclin E in normal and neoplastic cell cycles. Oncogene 2005; 24: 2776–2786.
Tuduri S, Crabbe L, Conti C, Tourriere H, Holtgreve-Grez H, Jauch A et al. Topoisomerase I suppresses genomic instability by preventing interference between replication and transcription. Nat Cell Biol 2009; 11: 1315–1324.
Gottipati P, Cassel TN, Savolainen L, Helleday T . Transcription-associated recombination is dependent on replication in mammalian cells. Mol Cell Biol 2008; 28: 154–164.
Gan W, Guan Z, Liu J, Gui T, Shen K, Manley JL et al. R-loop-mediated genomic instability is caused by impairment of replication fork progression. Genes Dev 2011; 25: 2041–2056.
Helmrich A, Ballarino M, Tora L . Collisions between replication and transcription complexes cause common fragile site instability at the longest human genes. Mol Cell 2011; 44: 966–977.
Akli S, Van Pelt CS, Bui T, Multani AS, Chang S, Johnson D et al. Overexpression of the low molecular weight cyclin E in transgenic mice induces metastatic mammary carcinomas through the disruption of the ARF-p53 pathway. Cancer Res 2007; 67: 7212–7222.
Resnitzky D, Gossen M, Bujard H, Reed SI . Acceleration of the G1/S phase transition by expression of cyclins D1 and E with an inducible system. Mol Cell Biol 1994; 14: 1669–1679.
Santoni-Rugiu E, Falck J, Mailand N, Bartek J, Lukas J . Involvement of Myc activity in a G(1)/S-promoting mechanism parallel to the pRb/E2F pathway. Mol Cell Biol 2000; 20: 3497–3509.
Ekholm-Reed S, Mendez J, Tedesco D, Zetterberg A, Stillman B, Reed SI . Deregulation of cyclin E in human cells interferes with prereplication complex assembly. J Cell Biol 2004; 165: 789–800.
Spruck CH, Won KA, Reed SI . Deregulated cyclin E induces chromosome instability. Nature 1999; 401: 297–300.
Rajagopalan H, Jallepalli PV, Rago C, Velculescu VE, Kinzler KW, Vogelstein B et al. Inactivation of hCDC4 can cause chromosomal instability. Nature 2004; 428: 77–81.
Keyomarsi K, Pardee AB . Redundant cyclin overexpression and gene amplification in breast cancer cells. Proc Natl Acad Sci USA 1993; 90: 1112–1116.
Thomson AM, Gillespie PJ, Blow JJ . Replication factory activation can be decoupled from the replication timing program by modulating Cdk levels. J Cell Biol 2010; 188: 209–221.
Lorvellec M, Dumon S, Maya-Mendoza A, Jackson D, Frampton J, Garcia P . B-Myb is critical for proper DNA duplication during an unperturbed S phase in mouse embryonic stem cells. Stem Cells 2010; 28: 1751–1759.
Petermann E, Maya-Mendoza A, Zachos G, Gillespie DA, Jackson DA, Caldecott KW . Chk1 requirement for high global rates of replication fork progression during normal vertebrate S phase. Mol Cell Biol 2006; 26: 3319–3326.
Arnaudeau C, Lundin C, Helleday T . DNA double-strand breaks associated with replication forks are predominantly repaired by homologous recombination involving an exchange mechanism in mammalian cells. J Mol Biol 2001; 307: 1235–1245.
Lundin C, Erixon K, Arnaudeau C, Schultz N, Jenssen D, Meuth M et al. Different roles for nonhomologous end joining and homologous recombination following replication arrest in mammalian cells. Mol Cell Biol 2002; 22: 5869–5878.
Montagnoli A, Valsasina B, Croci V, Menichincheri M, Rainoldi S, Marchesi V et al. A Cdc7 kinase inhibitor restricts initiation of DNA replication and has antitumor activity. Nat Chem Biol 2008; 4: 357–365.
Nimonkar AV, Genschel J, Kinoshita E, Polaczek P, Campbell JL, Wyman C et al. BLM-DNA2-RPA-MRN and EXO1-BLM-RPA-MRN constitute two DNA end resection machineries for human DNA break repair. Genes Dev 2011; 25: 350–362.
Sartori AA, Lukas C, Coates J, Mistrik M, Fu S, Bartek J et al. Human CtIP promotes DNA end resection. Nature 2007; 450: 509–514.
Jackson DA . S-phase progression in synchronized human cells. Exp Cell Res 1995; 220: 62–70.
Ge XQ, Jackson DA, Blow JJ . Dormant origins licensed by excess Mcm2-7 are required for human cells to survive replicative stress. Genes Dev 2007; 21: 3331–3341.
Courbet S, Gay S, Arnoult N, Wronka G, Anglana M, Brison O et al. Replication fork movement sets chromatin loop size and origin choice in mammalian cells. Nature 2008; 455: 557–560.
Geng Y, Lee Y-M, Welcker M, Swanger J, Zagozdzon A, Winer JD et al. Kinase-independent function of cyclin E. Mol Cell 2007; 25: 127–139.
Kawabata T, Luebben SW, Yamaguchi S, Ilves I, Matise I, Buske T et al. Stalled fork rescue via dormant replication origins in unchallenged S phase promotes proper chromosome segregation and tumor suppression. Mol Cell 2011; 41: 543–553.
Dereli-Oz A, Versini G, Halazonetis TD . Studies of genomic copy number changes in human cancers reveal signatures of DNA replication stress. Mol Oncol 2011; 5: 308–314.
Lukas C, Savic V, Bekker-Jensen S, Doil C, Neumann B, Pedersen RS et al. 53BP1 nuclear bodies form around DNA lesions generated by mitotic transmission of chromosomes under replication stress. Nat Cell Biol 2011; 13: 243–253.
Tuduri S, Crabbe L, Tourriere H, Coquelle A, Pasero P . Does interference between replication and transcription contribute to genomic instability in cancer cells? Cell Cycle 2010; 9: 1886–1892.
Zlatanou A, Despras E, Braz-Petta T, Boubakour-Azzouz I, Pouvelle C, Stewart GS et al. The hMsh2-hMsh6 complex acts in concert with monoubiquitinated PCNA and Pol eta in response to oxidative DNA damage in human cells. Mol Cell 2011; 43: 649–662.
Mailand N, Diffley JF . CDKs promote DNA replication origin licensing in human cells by protecting Cdc6 from APC/C-dependent proteolysis. Cell 2005; 122: 915–926.
Garcia P, Frampton J, Ballester A, Cales C . Ectopic expression of cyclin E allows non-endomitotic megakaryoblastic K562 cells to establish re-replication cycles. Oncogene 2000; 19: 1820–1833.
Acknowledgements
OM is supported by an EMBO Long-Term Fellowship. We thank Cancer Research UK for supporting this work financially, Dr Anastasia Zlatanou for cells and materials and Dr Agnieszka Gambus for helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Jones, R., Mortusewicz, O., Afzal, I. et al. Increased replication initiation and conflicts with transcription underlie Cyclin E-induced replication stress. Oncogene 32, 3744–3753 (2013). https://doi.org/10.1038/onc.2012.387
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.387
Keywords
This article is cited by
-
Oncogenic c-Myc induces replication stress by increasing cohesins chromatin occupancy in a CTCF-dependent manner
Nature Communications (2024)
-
cGAS-STING pathway expression correlates with genomic instability and immune cell infiltration in breast cancer
npj Breast Cancer (2024)
-
NUDT22 promotes cancer growth through pyrimidine salvage
Oncogene (2023)
-
Ring finger protein 126 promotes breast cancer metastasis and serves as a potential target to improve the therapeutic sensitivity of ATR inhibitors
Breast Cancer Research (2022)
-
Low-molecular-weight cyclin E deregulates DNA replication and damage repair to promote genomic instability in breast cancer
Oncogene (2022)