Abstract
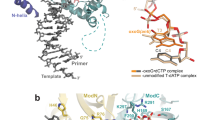
The 1,N6-ethenodeoxyadenosine (εdA) lesion is promutagenic and has been implicated in carcinogenesis. We show here that human Polι, a Y-family DNA polymerase, can promote replication through this lesion by proficiently incorporating a nucleotide opposite it. The structural basis of this action is rotation of the εdA adduct to the syn conformation in the Polι active site and presentation of its 'Hoogsteen edge' for hydrogen-bonding with incoming dTTP or dCTP. We also show that Polζ carries out the subsequent extension reaction and that efficiency of extension from εdA·T is notably higher than from εdA·C. Together, our studies reveal for the first time how the exocyclic εdA adduct is accommodated in a DNA polymerase active site, and they show that the combined action of Polι and Polζ provides for efficient and error-free synthesis through this potentially carcinogenic DNA lesion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bartsch, H., Barbin, A., Marion, M.J., Nair, J. & Guichard, Y. Formation, detection, and role in carcinogenesis of ethenobases in DNA. Drug Metab. Rev. 26, 349–371 (1994).
Chung, F.L., Zhang, L., Ocando, J.E. & Nath, R.G. Role of 1,N2-propanodeoxyguanosine adducts as endogenous DNA lesions in rodents and humans. IARC Sci. Publ. 150, 45–54 (1999).
Luczaj, W. & Skrzydlewska, E. DNA damage caused by lipid peroxidation products. Cell. Mol. Biol. Lett. 8, 391–413 (2003).
Levine, R.L. et al. Translesion DNA synthesis catalyzed by human pol eta and pol kappa across 1,N6-ethenodeoxyadenosine. J. Biol. Chem. 276, 18717–18721 (2001).
Prakash, S., Johnson, R.E. & Prakash, L. Eukaryotic translesion synthesis DNA polymerases: specificity of structure and function. Annu. Rev. Biochem. 74, 317–353 (2005).
Haracska, L. et al. Targeting of human DNA polymerase iota to the replication machinery via interaction with PCNA. Proc. Natl. Acad. Sci. USA 98, 14256–14261 (2001).
Johnson, R.E., Washington, M.T., Haracska, L., Prakash, S. & Prakash, L. Eukaryotic polymerases iota and zeta act sequentially to bypass DNA lesions. Nature 406, 1015–1019 (2000).
Tissier, A., McDonald, J.P., Frank, E.G. & Woodgate, R. poliota, a remarkably error-prone human DNA polymerase. Genes Dev. 14, 1642–1650 (2000).
Washington, M.T., Johnson, R.E., Prakash, L. & Prakash, S. Human DNA polymerase iota utilizes different nucleotide incorporation mechanisms dependent upon the template base. Mol. Cell. Biol. 24, 936–943 (2004).
Zhang, Y., Yuan, F., Wu, X. & Wang, Z. Preferential incorporation of G opposite template T by the low-fidelity human DNA polymerase iota. Mol. Cell. Biol. 20, 7099–7108 (2000).
Nair, D.T., Johnson, R.E., Prakash, S., Prakash, L. & Aggarwal, A.K. Replication by human DNA polymerase-iota occurs by Hoogsteen base-pairing. Nature 430, 377–380 (2004).
Nair, D.T., Johnson, R.E., Prakash, L., Prakash, S. & Aggarwal, A.K. Human DNA polymerase iota incorporates dCTP opposite template G via a G.C+ Hoogsteen base pair. Structure 13, 1569–1577 (2005).
Trincao, J. et al. Structure of the catalytic core of S. cerevisiae DNA polymerase eta: implications for translesion DNA synthesis. Mol. Cell 8, 417–426 (2001).
Zhou, B.L., Pata, J.D. & Steitz, T.A. Crystal structure of a DinB lesion bypass DNA polymerase catalytic fragment reveals a classic polymerase catalytic domain. Mol. Cell 8, 427–437 (2001).
Silvian, L.F., Toth, E.A., Pham, P., Goodman, M.F. & Ellenberger, T. Crystal structure of a DinB family error-prone DNA polymerase from Sulfolobus solfataricus. Nat. Struct. Biol. 8, 984–989 (2001).
Ling, H., Boudsocq, F., Woodgate, R. & Yang, W. Crystal structure of a Y-family DNA polymerase in action: a mechanism for error-prone and lesion-bypass replication. Cell 107, 91–102 (2001).
Uljon, S.N. et al. Crystal structure of the catalytic core of human DNA polymerase kappa. Structure 12, 1395–1404 (2004).
Nair, D.T., Johnson, R.E., Prakash, L., Prakash, S. & Aggarwal, A.K. Rev1 employs a novel mechanism of DNA synthesis using a protein template. Science 309, 2219–2222 (2005).
de los Santos, C. et al. NMR studies of 1,N6-ethenodeoxyadenosine adduct (epsilon dA) opposite deoxyguanosine in a DNA duplex. Epsilon dA(syn).dG(anti) pairing at the lesion site. Biochemistry 30, 1828–1835 (1991).
Dosanjh, M.K. et al. All four known cyclic adducts formed in DNA by the vinyl chloride metabolite chloroacetaldehyde are released by a human DNA glycosylase. Proc. Natl. Acad. Sci. USA 91, 1024–1028 (1994).
Saparbaev, M., Kleibl, K. & Laval, J. Escherichia coli, Saccharomyces cerevisiae, rat and human 3-methyladenine DNA glycosylases repair 1,N6-ethenoadenine when present in DNA. Nucleic Acids Res. 23, 3750–3755 (1995).
Eichman, B.F., O'Rourke, E.J., Radicella, J.P. & Ellenberger, T. Crystal structures of 3-methyladenine DNA glycosylase MagIII and the recognition of alkylated bases. EMBO J. 22, 4898–4909 (2003).
Johnson, R.E., Prakash, L. & Prakash, S. Biochemical evidence for the requirement of Hoogsteen base pairing for replication by human DNA polymerase iota. Proc. Natl. Acad. Sci. USA 102, 10466–10471 (2005).
Nair, D.T., Johnson, R.E., Prakash, S., Prakash, L. & Aggarwal, A.K. An incoming nucleotide imposes an anti to syn conformational change on the templating purine in the human DNA polymerase-iota active site. Structure 14, 749–755 (2006).
Asensio, J.L., Lane, A.N., Dhesi, J., Bergqvist, S. & Brown, T. The contribution of cytosine protonation to the stability of parallel DNA triple helices. J. Mol. Biol. 275, 811–822 (1998).
Plum, G.E. & Breslauer, K.J. Thermodynamics of an intramolecular DNA triple helix: a calorimetric and spectroscopic study of the pH and salt dependence of thermally induced structural transitions. J. Mol. Biol. 248, 679–695 (1995).
Kang, C.H. et al. Crystal structure of intercalated four-stranded d(C3T) at 1.4 A resolution. Proc. Natl. Acad. Sci. USA 91, 11636–11640 (1994).
Betts, L., Josey, J.A., Veal, J.M. & Jordan, S.R. A nucleic acid triple helix formed by a peptide nucleic acid-DNA complex. Science 270, 1838–1841 (1995).
Nunn, C.M., Trent, J.O. & Neidle, S. A model for the [C+-GxC]n triple helix derived from observation of the C+-GxC base triplet in a crystal structure. FEBS Lett. 416, 86–89 (1997).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Navaza, J. AMoRe: an automated package for molecular replacement. Acta Crystallogr. 50, 157–163 (1994).
Brunger, A.T. et al. Crystallography & NMR system: a software suite for macromolecular structure determination. Acta Crystallogr. D Biol.Crystallogr. 54, 905 (1998).
Jones, A.T., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Winn, M.D., Murshudov, G.N. & Papiz, M.Z. Macromolecular TLS refinement in REFMAC at moderate resolutions. Methods Enzymol. 374, 300–321 (2003).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. A 47, 110–119 (1993).
Acknowledgements
We thank the staff at the Advanced Photon Source (beamline 19ID) and Brookhaven National Laboratory (beamline X29) for facilitating X-ray data collection. We thank S. Townson and S. Lone for help with data collection and DNA purification. This work was supported by grant CA115856 from the US National Institutes of Health (S.P. and A.K.A.).
Author information
Authors and Affiliations
Contributions
D.T.N. performed the crystallographic experiments, R.E.J. conducted the kinetic experiments and all of the authors contributed to the concepts and to the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nair, D., Johnson, R., Prakash, L. et al. Hoogsteen base pair formation promotes synthesis opposite the 1,N6-ethenodeoxyadenosine lesion by human DNA polymerase ι. Nat Struct Mol Biol 13, 619–625 (2006). https://doi.org/10.1038/nsmb1118
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1118
This article is cited by
-
Etheno adducts: from tRNA modifications to DNA adducts and back to miscoding ribonucleotides
Genes and Environment (2021)
-
Structure and function relationships in mammalian DNA polymerases
Cellular and Molecular Life Sciences (2020)
-
Mechanism of error-free DNA synthesis across N1-methyl-deoxyadenosine by human DNA polymerase-ι
Scientific Reports (2017)
-
Identification of amino acid residues involved in the dRP-lyase activity of human Pol ι
Scientific Reports (2017)
-
Comparable Stability of Hoogsteen and Watson–Crick Base Pairs in Ionic Liquid Choline Dihydrogen Phosphate
Scientific Reports (2014)