Abstract
Effectors secreted by the type III secretion system are essential for bacterial pathogenesis. Members of the Yersinia outer-protein J (YopJ) family of effectors found in diverse plant and animal pathogens depend on a protease-like catalytic triad to acetylate host proteins and produce virulence. However, the structural basis for this noncanonical acetyltransferase activity remains unknown. Here, we report the crystal structures of the YopJ effector HopZ1a, produced by the phytopathogen Pseudomonas syringae, in complex with the eukaryote-specific cofactor inositol hexakisphosphate (IP6) and/or coenzyme A (CoA). Structural, computational and functional characterizations reveal a catalytic core with a fold resembling that of ubiquitin-like cysteine proteases and an acetyl-CoA-binding pocket formed after IP6-induced structural rearrangements. Modeling-guided mutagenesis further identified key IP6-interacting residues of Salmonella effector AvrA that are required for acetylating its substrate. Our study reveals the structural basis of a novel class of acetyltransferases and the conserved allosteric regulation of YopJ effectors by IP6.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galán, J.E., Lara-Tejero, M., Marlovits, T.C. & Wagner, S. Bacterial type III secretion systems: specialized nanomachines for protein delivery into target cells. Annu. Rev. Microbiol. 68, 415–438 (2014).
Dean, P. Functional domains and motifs of bacterial type III effector proteins and their roles in infection. FEMS Microbiol. Rev. 35, 1100–1125 (2011).
Ma, W., Dong, F.F.T., Stavrinides, J. & Guttman, D.S. Type III effector diversification via both pathoadaptation and horizontal transfer in response to a coevolutionary arms race. PLoS Genet. 2, e209 (2006).
Orth, K. et al. Inhibition of the mitogen-activated protein kinase kinase superfamily by a Yersinia effector. Science 285, 1920–1923 (1999).
Mukherjee, S. et al. Yersinia YopJ acetylates and inhibits kinase activation by blocking phosphorylation. Science 312, 1211–1214 (2006).
Trosky, J.E. et al. VopA inhibits ATP binding by acetylating the catalytic loop of MAPK kinases. J. Biol. Chem. 282, 34299–34305 (2007).
Jones, R.M. et al. Salmonella AvrA coordinates suppression of host immune and apoptotic defenses via JNK pathway blockade. Cell Host Microbe 3, 233–244 (2008).
Lee, A.H. et al. A bacterial acetyltransferase destroys plant microtubule networks and blocks secretion. PLoS Pathog. 8, e1002523 (2012).
Jiang, S. et al. Bacterial effector activates jasmonate signaling by directly targeting JAZ transcriptional repressors. PLoS Pathog. 9, e1003715 (2013).
Tasset, C. et al. Autoacetylation of the Ralstonia solanacearum effector PopP2 targets a lysine residue essential for RRS1-R-mediated immunity in Arabidopsis. PLoS Pathog. 6, e1001202 (2010).
Le Roux, C. et al. A receptor pair with an integrated decoy converts pathogen disabling of transcription factors to immunity. Cell 161, 1074–1088 (2015).
Sarris, P.F. et al. A plant immune receptor detects pathogen effectors that target WRKY transcription factors. Cell 161, 1089–1100 (2015).
Lee, J. et al. Acetylation of an NB-LRR plant immune-effector complex suppresses immunity. Cell Rep. 13, 1670–1682 (2015).
Cheong, M.S. et al. AvrBsT acetylates Arabidopsis ACIP1, a protein that associates with microtubules and is required for immunity. PLoS Pathog. 10, e1003952 (2014).
Mittal, R., Peak-Chew, S.Y., Sade, R.S., Vallis, Y. & McMahon, H.T. The acetyltransferase activity of the bacterial toxin YopJ of Yersinia is activated by eukaryotic host cell inositol hexakisphosphate. J. Biol. Chem. 285, 19927–19934 (2010).
Ma, K.W. et al. Two serine residues in Pseudomonas syringae effector HopZ1a are required for acetyltransferase activity and association with the host co-factor. New Phytol. 208, 1157–1168 (2015).
Choudhary, C., Weinert, B.T., Nishida, Y., Verdin, E. & Mann, M. The growing landscape of lysine acetylation links metabolism and cell signalling. Nat. Rev. Mol. Cell Biol. 15, 536–550 (2014).
Orth, K. et al. Disruption of signaling by Yersinia effector YopJ, a ubiquitin-like protein protease. Science 290, 1594–1597 (2000).
Mossessova, E. & Lima, C.D. Ulp1-SUMO crystal structure and genetic analysis reveal conserved interactions and a regulatory element essential for cell growth in yeast. Mol. Cell 5, 865–876 (2000).
Holm, L. & Rosenström, P. Dali server: conservation mapping in 3D. Nucleic Acids Res. 38, W545–W549 (2010).
Vetting, M.W. et al. Structure and functions of the GNAT superfamily of acetyltransferases. Arch. Biochem. Biophys. 433, 212–226 (2005).
Utley, R.T. & Côté, J. The MYST family of histone acetyltransferases. Curr. Top. Microbiol. Immunol. 274, 203–236 (2003).
Mukherjee, S., Hao, Y.H. & Orth, K. A newly discovered post-translational modification: the acetylation of serine and threonine residues. Trends Biochem. Sci. 32, 210–216 (2007).
Lupardus, P.J., Shen, A., Bogyo, M. & Garcia, K.C. Small molecule-induced allosteric activation of the Vibrio cholerae RTX cysteine protease domain. Science 322, 265–268 (2008).
Pruitt, R.N. et al. Structure-function analysis of inositol hexakisphosphate-induced autoprocessing in Clostridium difficile toxin A. J. Biol. Chem. 284, 21934–21940 (2009).
Hari, S.B., Merritt, E.A. & Maly, D.J. Sequence determinants of a specific inactive protein kinase conformation. Chem. Biol. 20, 806–815 (2013).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Adams, P.D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D Biol. Crystallogr. 58, 1948–1954 (2002).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Cunnac, S. et al. Genetic disassembly and combinatorial reassembly identify a minimal functional repertoire of type III effectors in Pseudomonas syringae. Proc. Natl. Acad. Sci. USA 108, 2975–2980 (2011).
Morgan, R.L. et al. Catalytic domain of the diversified Pseudomonas syringae type III effector HopZ1 determines the allelic specificity in plant hosts. Mol. Microbiol. 76, 437–455 (2010).
Micsonai, A. et al. Accurate secondary structure prediction and fold recognition for circular dichroism spectroscopy. Proc. Natl. Acad. Sci. USA 112, E3095–E3103 (2015).
Gille, C., Fähling, M., Weyand, B., Wieland, T. & Gille, A. Alignment-Annotator web server: rendering and annotating sequence alignments. Nucleic Acids Res. 42, W3–W6 (2014).
Søndergaard, C.R., Olsson, M.H.M., Rostkowski, M. & Jensen, J.H. Improved treatment of ligands and coupling effects in empirical calculation and rationalization of pKa values. J. Chem. Theory Comput. 7, 2284–2295 (2011).
Klauda, J.B. et al. Update of the CHARMM all-atom additive force field for lipids: validation on six lipid types. J. Phys. Chem. B 114, 7830–7843 (2010).
Vanommeslaeghe, K., Raman, E.P. & MacKerell, A.D. Jr. Automation of the CHARMM General Force Field (CGenFF) II: assignment of bonded parameters and partial atomic charges. J. Chem. Inf. Model. 52, 3155–3168 (2012).
Schmauder, R., Kosanic, D., Hovius, R. & Vogel, H. Correlated optical and electrical single-molecule measurements reveal conformational diffusion from ligand binding to channel gating in the nicotinic acetylcholine receptor. ChemBioChem 12, 2431–2434 (2011).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38, 27–28 (1996).
Pronk, S. et al. GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 29, 845–854 (2013).
Skjærven, L., Yao, X.Q., Scarabelli, G. & Grant, B.J. Integrating protein structural dynamics and evolutionary analysis with Bio3D. BMC Bioinformatics 15, 399 (2014).
Grant, B.J., Rodrigues, A.P., ElSawy, K.M., McCammon, J.A. & Caves, L.S. Bio3d: an R package for the comparative analysis of protein structures. Bioinformatics 22, 2695–2696 (2006).
Scarabelli, G. & Grant, B.J. Kinesin-5 allosteric inhibitors uncouple the dynamics of nucleotide, microtubule, and neck-linker binding sites. Biophys. J. 107, 2204–2213 (2014).
Van Wart, A.T., Durrant, J., Votapka, L. & Amaro, R.E. Weighted implementation of suboptimal paths (WISP): an optimized algorithm and tool for dynamical network analysis. J. Chem. Theory Comput. 10, 511–517 (2014).
Bakan, A., Meireles, L.M. & Bahar, I. ProDy: protein dynamics inferred from theory and experiments. Bioinformatics 27, 1575–1577 (2011).
Acknowledgements
This work was supported by funds from a Kimmel Scholar Award from the Sidney Kimmel Foundation for Cancer Research, a University of California Cancer Research Coordination Committee Award (CRC-15-380558) and the March of Dimes Foundation (1-FY15-345) to J.S., and grants from the US NSF (IOS no. 0847870) and the USDA Agriculture Experimental Station Funding (CA-R-PPA-5075-H) to W.M. We thank A. Collmer (Cornell University) for providing the P. syringae strain PtoD28E, D. Maly (University of Washington) for the MKK4 construct and D. Borchardt (UC Riverside) for technical support in NMR and CD spectroscopy.
Author information
Authors and Affiliations
Contributions
Z.-M.Z. determined the crystal structures of HopZ1a complexes and conducted ITC assays. K.-W.M. characterized NMR, CD and fluorescence spectra of HopZ1a or AvrA proteins. K.-W.M., S.J. and E.H. performed in vitro acetylation assays and in vivo functional analyses. S.Y. performed computational analysis. Z.-M.Z. and Y.L. crystalized HopZ1a complexes. Z.-M.Z. and S.P. performed limited proteolysis and mass spectrometry analysis. W.M. and J.S. designed and organized the study, Z.-M.Z., K.-W.M., S.Y., W.M. and J.S. prepared the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Binding of IP6 and CoA to HopZ1a.
(a) Crystal structure of the HopZ1a–IP6 complex. IP6 is shown in the ball-and-stick model. (b) A close-up view of the catalytic center of HopZ1a. CoA is shown in the ball-and-stick model. Hydrogen bonds are shown in red dashed line. (c) An alternative conformation of H150 in the HopZ1a–IP6–CoA structure due to crystal packing. Citrate in the packing buffer is shown in stick representation. (d) Surface electrostatic view of HopZ1a showing the binding pocket of IP6. IP6 is shown in stick representation. (e) Fo-Fc omit map of IP6 in the HopZ1a–IP6-CoA complex structure at a contour level of 1.5σ. (f) Surface electrostatic view of HopZ1a showing the binding pocket of CoA. CoA is shown in stick representation. (g) Fo-Fc omit map of CoA in the HopZ1a–IP6–CoA complex structure at a contour level of 1.5σ.
Supplementary Figure 2 Sequence alignment of YopJ family effectors and ULP1.
Sequence alignment of YopJ family effectors with demonstrated acetyltransferase activity (HopZ1a and HopZ3 from Pseudomonas syringae, AvrBsT from Xanthomonas campestris, PopP2 from Ralstonia solanacearum, VopA from Vibrio parahaemolyticus, AvrA from Salmonella enterica, and YopJ from Yersinia pestis) and ULP1. Secondary structure elements of HopZ1a are shown above the aligned sequences. The catalytic triads are marked by red triangles; residues involved in IP6 binding are marked by green diamonds; residues involved in CoA binding are marked by black triangles; and the region in ULP1 that is involved in SUMO binding is underlined.
Supplementary Figure 3 In planta activity analyses of HopZ1a mutants.
(a) IP6- and CoA-binding mutants of HopZ1a-HA were expressed in PtoD28E and the transformants were used to inoculate leaves of 5-week-old Arabidopsis eco. Col-0. Electrolyte leakage of the inoculated leaf disks was measured during a time course. Error bars, S.D., n=3 leaf disks. (b) Immunoblot showing the expression of wild-type and mutant HopZ1a-HA in PtoD28E. HopZ1a proteins were induced by growing P. syringae in M9 minimal medium and detected by anti-HA antibody (Roche Diagnostics). PtoD28E carrying the empty vector (EV) was used as a negative control. (c) Effect of single mutations of the three AcCoA-binding residues on the HR-triggering ability of HopZ1a in Arabidopsis eco. Col-0. The catalytic mutant C216A was used as a control. Leaves showing HR were labeled with an asterisk. Ratio: number of leaves with indicated phenotype/total number of leaves inoculated in each treatment.
Supplementary Figure 4 1D 1H NMR and CD spectra of wild-type HopZ1a, the catalytic mutant and the IP6-binding-deficient mutants.
(a-e, left and middle) Selected regions of NMR spectra and CD spectra in the absence (black) or presence (red) of IP6 for WT. Note that the NMR signal with 1H chemical shift of -0.7ppm (marked with asterisk) only appears in the NMR spectra of wild-type and C216A HopZ1a when mixed with IP6. (a-e, right) Far-UV CD spectra in the presence of various amount of IP6 are overlaid. A slight but notable decrease in ellipticities at 208 nm and 222 nm of wild-type HopZ1a or the catalytic mutant C216A upon addition of IP6 indicated increases of helical content.
Supplementary Figure 5 Elastase cleavage assay of HopZ1a in the presence or absence of IP6 and/or CoA.
Thirty µg of HopZ1a was digested in a 30 µL reaction mixture incubated on ice for 1 hour. SDS-PAGE was used to separate the elastase cleavage products of HopZ1a.
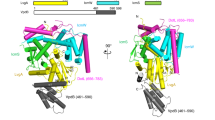
Supplementary Figure 6 Molecular dynamics simulation of HopZ1a in IP6 and/or CoA binding states.
(a) The B-factor from MD simulations of HopZ1a, its IP6 bound form and/or IP6–CoA bound form. Regions with high B-factor are indicated by red putty. Low B-factor regions are depicted by blue putty. (b) Simultaneous view of community residue interaction network and 3D structure of HopZ1a (left), HopZ1a–IP6 (middle) and HopZ1a–IP6–CoA (right). The correlation network representation nodes correspond to protein residues connected by edges and weighted by the strength of their respective correlation values. Variations in the connectivity of the network give rise to modules or local communities in the network. Nodes belonging to the same community are more strongly and densely interconnected to one another with weaker connections to other nodes in the network43.
Supplementary Figure 7 Conserved enzymatic regulation of YopJ effectors.
(a) Three-dimensional sequence alignment of IP6–binding (left) and CoA-binding residues (right) in YopJ family effectors. (b) Structural modeling of AvrA based on the structure of HopZ1a–IP6. The IP6-binding pocket is boxed and amplified. (c) Conformational changes of wild-type and mutant AvrA upon the addition of IP6 (protein:IP6 molar ratio = 1:2) were determined by tryptophan fluorescence analysis over time.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1–3 (PDF 1902 kb)
Supplementary Data Set 1
Uncropped images for Figs. 2–4 (PDF 495 kb)
Normal mode analysis (NMA) of HopZ1a upon IP6 binding
The regulatory and catalytic domains are colored in pink and cyan, respectively. The NMA analysis indicates that the movements loopαL-αM and loopβ7-β8 correlate with each other after the binding of IP6 molecule. The length of arrow correlates with the motion of each domain. (AVI 4967 kb)
Source data
Rights and permissions
About this article
Cite this article
Zhang, ZM., Ma, KW., Yuan, S. et al. Structure of a pathogen effector reveals the enzymatic mechanism of a novel acetyltransferase family. Nat Struct Mol Biol 23, 847–852 (2016). https://doi.org/10.1038/nsmb.3279
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3279
This article is cited by
-
Acetylproteomics analyses reveal critical features of lysine-ε-acetylation in Arabidopsis and a role of 14-3-3 protein acetylation in alkaline response
Stress Biology (2022)
-
Secondary-structure switch regulates the substrate binding of a YopJ family acetyltransferase
Nature Communications (2021)
-
In-silico structural analysis of Pseudomonas syringae effector HopZ3 reveals ligand binding activity and virulence function
Journal of Plant Research (2021)
-
Bacterial virulence mediated by orthogonal post-translational modification
Nature Chemical Biology (2020)
-
A Chlamydia effector combining deubiquitination and acetylation activities induces Golgi fragmentation
Nature Microbiology (2018)