Key Points
-
Antibodies that recognize DNA (anti-DNA antibodies) can bind to sites on the phosphodiester backbone of single-stranded DNA and double-stranded DNA, to nucleotide sequences or to higher-order structures such as nucleosomes
-
The molecular properties of anti-DNA antibodies, as well as the associated genetic properties, including variable-region somatic mutations, point to a role for antigen selection in anti-DNA antibody generation
-
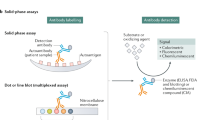
In the absence of a 'gold standard', various assay formats exist for anti-DNA antibody testing, differing in the nature of DNA substrates and the conditions for binding and detection of antibodies
-
High-affinity binding by an anti-DNA antibody depends on monogamous bivalency, in which both Fab sites of an IgG molecule contact the same polynucleotide chain
-
The use of anti-DNA antibody testing as a measure of disease activity to determine clinical trial eligibility depends on clear understanding of assay differences and the role of anti-DNA antibodies in pathogenesis
Abstract
Antibodies that recognize and bind to DNA (anti-DNA antibodies) are serological hallmarks of systemic lupus erythematosus (SLE) and key markers for diagnosis and disease activity. In addition to common use in the clinic, anti-DNA antibody testing now also determines eligibility for clinical trials, raising important questions about the nature of the antibody–antigen interaction. At present, no 'gold standard' for serological assessment exists, and anti-DNA antibody binding can be measured with a variety of assay formats, which differ in the nature of the DNA substrates and in the conditions for binding and detection of antibodies. A mechanism called monogamous bivalency — in which high avidity results from simultaneous interaction of IgG Fab sites with a single polynucleotide chain — determines anti-DNA antibody binding; this mechanism might affect antibody detection in different assay formats. Although anti-DNA antibodies can promote pathogenesis by depositing in the kidney or driving cytokine production, they are not all alike, pathologically, and anti-DNA antibody expression does not necessarily correlate with active disease. Levels of anti-DNA antibodies in patients with SLE can vary over time, distinguishing anti-DNA antibodies from other pathogenic antinuclear antibodies. Elucidation of the binding specificities and the pathogenic roles of anti-DNA antibodies in SLE should enable improvements in the design of informative assays for both clinical and research purposes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hahn, B. H. Antibodies to DNA. N. Engl. J. Med. 338, 1359–1368 (1998).
Jang, Y. J. & Stollar, B. D. Anti-DNA antibodies: aspects of structure and pathogenicity. Cell. Mol. Life Sci. 60, 309–320 (2003).
Tsokos, G. C. Systemic lupus erythematosus. N. Engl. J. Med. 365, 2110–2121 (2011).
Breden, F. et al. Comparison of antibody repertoires produced by HIV-1 infection, other chronic and acute infections, and systemic autoimmune disease. PLoS ONE 6, e16857 (2011).
Dörner, T., Giesecke, C. & Lipsky, P. E. Mechanisms of B cell autoimmunity in SLE. Arthritis Res. Ther. 13, 243 (2011).
Furie, R. et al. A phase III, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits B lymphocyte stimulator, in patients with systemic lupus erythematosus. Arthritis Rheum. 63, 3918–3930 (2011).
van Vollenhoven, R. F. et al. Belimumab in the treatment of systemic lupus erythematosus: high disease activity predictors of response. Ann. Rheum. Dis. 71, 1343–1349 (2012).
Egner, W. The use of laboratory tests in the diagnosis of SLE. J. Clin. Pathol. 53, 424–432 (2000).
Rekvig, O. P. The anti-DNA antibody: origin and impact, dogmas and controversies. Nat. Rev. Rheumatol. 11, 530–540 (2015).
Ceppellini, R., Polli, E. & Celada, F. A DNA-reacting factor in serum of a patient with lupus erythematosus diffusus. Proc. Soc. Exp. Biol. Med. 96, 572–574 (1957).
Miescher, P. & Strassle, R. New serological methods for the detection of the L.E. factor. Vox Sang. 2, 283–287 (1957).
Robbins, W. C., Holman, H. R., Deicher, H. & Kunkel, H. G. Complement fixation with cell nuclei and DNA in lupus erythematosus. Proc. Soc. Exp. Biol. Med. 96, 575–579 (1957).
Seligmann, M. [Demonstration in the blood of patients with disseminated lupus erythematosus a substance determining a precipitation reaction with desoxyribonucleic acid][French]. C. R. Hebd. Seances Acad. Sci. 245, 243–245 (1957).
Stollar, B. D. & Papalian, M. Secondary structure in denatured DNA is responsible for its reaction with antinative DNA antibodies of systemic lupus erythematosus sera. J. Clin. Invest. 66, 210–219 (1980).
Pisetsky, D. S. Standardization of anti-DNA antibody assays. Immunol. Res. 56, 420–424 (2013).
Stollar, B. D. The specificity and applications of antibodies to helical nucleic acids. CRC Crit. Rev. Biochem. 3, 45–69 (1975).
van Steensel, B. Chromatin: constructing the big picture. EMBO J. 30, 1885–1895 (2011).
Khorasanizadeh, S. The nucleosome: from genomic organization to genomic regulation. Cell 116, 259–272 (2004).
Choy, J. S. & Lee, T. H. Structural dynamics of nucleosomes at single-molecule resolution. Trends Biochem. Sci. 37, 425–435 (2012).
Grootscholten, C. et al. A prospective study of anti-chromatin and anti-C1q autoantibodies in patients with proliferative lupus nephritis treated with cyclophosphamide pulses or azathioprine/methylprednisolone. Ann. Rheum. Dis. 66, 693–696 (2007).
Bigler, C. et al. Antinucleosome antibodies as a marker of active proliferative lupus nephritis. Am. J. Kidney Dis. 51, 624–629 (2008).
Mehra, S. & Fritzler, M. J. The spectrum of anti-chromatin/nucleosome autoantibodies: independent and interdependent biomarkers of disease. J. Immunol. Res. 2014, 368274 (2014).
Rekvig, O. P., van der Vlag, J. & Seredkina, N. Review: antinucleosome antibodies: a critical reflection on their specificities and diagnostic impact. Arthritis Rheumatol. 66, 1061–1069 (2014).
Li, T. et al. Anti-nucleosome antibodies outperform traditional biomarkers as longitudinal indicators of disease activity in systemic lupus erythematosus. Rheumatology (Oxford) 54, 449–457 (2015).
Burlingame, R. W., Rubin, R. L., Balderas, R. S. & Theofilopoulos, A. N. Genesis and evolution of antichromatin autoantibodies in murine lupus implicates T-dependent immunization with self antigen. J. Clin. Invest. 91, 1687–1696 (1993).
Burlingame, R. W., Boey, M. L., Starkebaum, G. & Rubin, R. L. The central role of chromatin in autoimmune responses to histones and DNA in systemic lupus erythematosus. J. Clin. Invest. 94, 184–192 (1994).
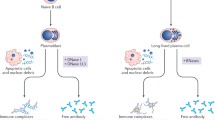
Jahr, S. et al. DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res. 61, 1659–1665 (2001).
Jiang, N., Reich, C. F. 3rd & Pisetsky, D. S. Role of macrophages in the generation of circulating blood nucleosomes from dead and dying cells. Blood 102, 2243–2250 (2003).
Tsang, J. C. & Lo, Y. M. Circulating nucleic acids in plasma/serum. Pathology 39, 197–207 (2007).
Schwarzenbach, H., Hoon, D. S. & Pantel, K. Cell-free nucleic acids as biomarkers in cancer patients. Nat. Rev. Cancer 11, 426–437 (2011).
Pisetsky, D. S. The origin and properties of extracellular DNA: from PAMP to DAMP. Clin. Immunol. 144, 32–40 (2012).
Pisetsky, D. S. The translocation of nuclear molecules during inflammation and cell death. Antioxid. Redox Signal. 20, 1117–1125 (2014).
Galluzzi, L. et al. Essential versus accessory aspects of cell death: recommendations of the NCCD 2015. Cell Death Differ. 22, 58–73 (2015).
Nyström, S. et al. TLR activation regulates damage-associated molecular pattern isoforms released during pyroptosis. EMBO J. 32, 86–99 (2013).
Yang, H., Antoine, D. J., Andersson, U. & Tracey, K. J. The many faces of HMGB1: molecular structure-functional activity in inflammation, apoptosis, and chemotaxis. J. Leukoc. Biol. 93, 865–873 (2013).
Pisetsky, D. S. The complex role of DNA, histones and HMGB1 in the pathogenesis of SLE. Autoimmunity 47, 487–493 (2014).
Villanueva, E. et al. Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J. Immunol. 187, 538–552 (2011).
Leffler, J. et al. Neutrophil extracellular traps that are not degraded in systemic lupus erythematosus activate complement exacerbating the disease. J. Immunol. 188, 3522–3531 (2012).
Atamaniuk, J. et al. Analysing cell-free plasma DNA and SLE disease activity. Eur. J. Clin. Invest. 41, 579–583 (2011).
Zhang, S. et al. Elevated plasma cfDNA may be associated with active lupus nephritis and partially attributed to abnormal regulation of neutrophil extracellular traps (NETs) in patients with systemic lupus erythematosus. Intern. Med. 53, 2763–2771 (2014).
Ullal, A. J. et al. Microparticles as antigenic targets of antibodies to DNA and nucleosomes in systemic lupus erythematosus. J. Autoimmun. 36, 173–180 (2011).
Nielsen, C. T. et al. Increased IgG on cell-derived plasma microparticles in systemic lupus erythematosus is associated with autoantibodies and complement activation. Arthritis Rheum. 64, 1227–1236 (2012).
Petri, M. et al. Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum. 64, 2677–2686 (2012).
Rekvig, O. P. Anti-dsDNA antibodies as a classification criterion and a diagnostic marker for systemic lupus erythematosus: critical remarks. Clin. Exp. Immunol. 179, 5–10 (2015).
Ward, M. M., Pisetsky, D. S. & Christenson, V. D. Antidouble stranded DNA antibody assays in systemic lupus erythematosus: correlations of longitudinal antibody measurements. J. Rheumatol. 16, 609–613 (1989).
Venner, A. A. et al. Comparison of three anti-dsDNA assays: performance and correlation with systemic lupus erythematosus disease activity. Clin. Biochem. 46, 317–320 (2013).
Enocsson, H. et al. Four anti-dsDNA antibody assays in relation to systemic lupus erythematosus disease specificity and activity. J. Rheumatol. 42, 817–825 (2015).
Arden, L. A., Lakmaker, F. & Feltkamp, T. E. Immunology of DNA. II. The effect of size and structure of the antigen on the Farr assay. J. Immunol. Methods 10, 39–48 (1976).
Hillebrand, J. J., Bernelot Moens, H. J. & Mulder, A. H. Changes in Farr radioimmunoassay and EliA fluorescence immunoassay anti-dsDNA in relation to exacerbation of SLE. Lupus 22, 1169–1173 (2013).
Crothers, D. M. & Metzger, H. The influence of polyvalency on the binding properties of antibodies. Immunochemistry 9, 341–357 (1972).
Romans, D. G., Tilley, C. A. & Dorrington, K. J. Monogamous bivalency of IgG antibodies. I. Deficiency of branched ABHI-active oligosaccharide chains on red cells of infants causes the weak antiglobulin reactions in hemolytic disease of the newborn due to ABO incompatibility. J. Immunol. 124, 2807–2811 (1980).
Kaufman, E. N. & Jain, R. K. Effect of bivalent interaction upon apparent antibody affinity: experimental confirmation of theory using fluorescence photobleaching and implications for antibody binding assays. Cancer Res. 52, 4157–4167 (1992).
Werner, T. C., Bunting, J. R. & Cathou, R. E. The shape of immunoglobulin G molecules in solution. Proc. Natl Acad. Sci. USA 69, 795–799 (1972).
Papalian, M., Lafer, E., Wong, R. & Stollar, B. D. Reaction of systemic lupus erythematosus antinative DNA antibodies with native DNA fragments from 20 to 1,200 base pairs. J. Clin. Invest. 65, 469–477 (1980).
Ali, R., Dersimonian, H. & Stollar, B. D. Binding of monoclonal anti-native DNA autoantibodies to DNA of varying size and conformation. Mol. Immunol. 22, 1415–1422 (1985).
Pisetsky, D. S. & Reich, C. F. The influence of DNA size on the binding of anti-DNA antibodies in the solid and fluid phase. Clin. Immunol. Immunopathol. 72, 350–356 (1994).
Lennek, R., Baldwin, A. S. Jr, Waller, S. J., Morley, K. W. & Taylor, R. P. Studies of the physical biochemistry and complement-fixing properties of DNA/anti-DNA immune complexes. J. Immunol. 127, 602–608 (1981).
Waller, S. J., Taylor, R. P., Wright, E. L., Morley, K. W. & Johns, M. DNA/anti-DNA complexes: correlation of size and complement fixation. Arthritis Rheum. 24, 651–657 (1981).
Radic, M. Z. et al. Residues that mediate DNA binding of autoimmune antibodies. J. Immunol. 150, 4966–4977 (1993).
Radic, M. Z. & Weigert, M. Genetic and structural evidence for antigen selection of anti-DNA antibodies. Ann. Rev. Immunol. 12, 487–520 (1994).
Li, Z., Schettino, E. W., Padlan, E. A., Ikematsu, H. & Casali, P. Structure-function analysis of a lupus anti-DNA autoantibody: central role of the heavy chain complementarity-determining region 3 Arg in binding of double- and single-stranded DNA. Eur. J. Immunol. 30, 2015–2026 (2000).
Zhang, J., Jacobi, A. M., Wang, T. & Diamond, B. Pathogenic autoantibodies in systemic lupus erythematosus are derived from both self-reactive and non-self-reactive B cells. Mol. Med. 14, 675–681 (2008).
Richardson, C. et al. Molecular basis of 9G4 B cell autoreactivity in human systemic lupus erythematosus. J. Immunol. 191, 4926–4939 (2013).
Schroeder, K., Herrmann, M. & Winkler, T. H. The role of somatic hypermutation in the generation of pathogenic antibodies in SLE. Autoimmunity 46, 121–127 (2013).
Mohan, C., Adams, S., Stanik, V. & Datta, S. K. Nucleosome: a major immunogen for pathogenic autoantibody-inducing T cells of lupus. J. Exp. Med. 177, 1367–1381 (1993).
Kaliyaperumal, A., Mohan, C., Wu, W. & Datta, S. K. Nucleosomal peptide epitopes for nephritis-inducing T helper cells of murine lupus. J. Exp. Med. 183, 2459–2469 (1996).
Sano, H. & Morimoto, C. DNA isolated from DNA/anti-DNA antibody immune complexes in systemic lupus erythematosus is rich in guanine-cytosine content. J. Immunol. 128, 1341–1345 (1982).
Rumore, P. M. & Steinman, C. R. Endogenous circulating DNA in systemic lupus erythematosus. Occurrence as multimeric complexes bound to histone. J. Clin. Invest. 86, 69–74 (1990).
Chan, R. W. et al. Plasma DNA aberrations in systemic lupus erythematosus revealed by genomic and methylomic sequencing. Proc. Natl Acad. Sci. USA 111, E5302–E5311 (2014).
Sano, H. et al. Binding properties of human anti-DNA antibodies to cloned human DNA fragments. Scand. J. Immunol. 30, 51–63 (1989).
Uccellini, M. B., Busto, P., Debatis, M., Marshak-Rothstein, A. & Viglianti, G. A. Selective binding of anti-DNA antibodies to native dsDNA fragments of differing sequence. Immunol. Lett. 143, 85–91 (2012).
Karounos, D. G., Grudier, J. P. & Pisetsky, D. S. Spontaneous expression of antibodies to DNA of various species origin in sera of normal subjects and patients with systemic lupus erythematosus. J. Immunol. 140, 451–455 (1988).
Robertson, C. R., Gilkeson, G. S., Ward, M. M. & Pisetsky, D. S. Patterns of heavy and light chain utilization in the antibody response to single-stranded bacterial DNA in normal human subjects and patients with systemic lupus erythematosus. Clin. Immunol. Immunopathol. 62, 25–32 (1992).
Bunyard, M. P. & Pisetsky, D. S. Characterization of antibodies to bacterial double-stranded DNA in the sera of normal human subjects. Int. Arch. Allergy Immunol. 105, 122–127 (1994).
Fredriksen, K., Skogsholm, A., Flaegstad, T., Traavik, T. & Rekvig, O. P. Antibodies to dsDNA are produced during primary BK virus infection in man, indicating that anti-dsDNA antibodies may be related to virus replication in vivo. Scand. J. Immunol. 38, 401–406 (1993).
Fredriksen, K., Osei, A., Sundsfjord, A., Traavik, T. & Rekvig, O. P. On the biological origin of anti-double-stranded (ds) DNA antibodies: systemic lupus erythematosus-related anti-dsDNA antibodies are induced by polyomavirus BK in lupus-prone (NZBxNZW) F1 hybrids, but not in normal mice. Eur. J. Immunol. 24, 66–70 (1994).
Pisetsky, D. S. & Drayton, D. M. Deficient expression of antibodies specific for bacterial DNA by patients with systemic lupus erythematosus. Proc. Assoc. Am. Physicians 109, 237–244 (1997).
Hamilton, K. J., Schett, G., Reich, C. F. 3rd, Smolen, J. S. & Pisetsky, D. S. The binding of sera of patients with SLE to bacterial and mammalian DNA. Clin. Immunol. 118, 209–218 (2006).
Wang, X., Stearns, N. A., Li, X. & Pisetsky, D. S. The effect of polyamines on the binding of anti-DNA antibodies from patients with SLE and normal human subjects. Clin. Immunol. 153, 94–103 (2014).
Koffler, D., Agnello, V., Thoburn, R. & Kunkel, H. G. Systemic lupus erythematosus: prototype of immune complex nephritis in man. J. Exp. Med. 134, 169–179 (1971).
Bruneau, C. & Benveniste, J. Circulating DNA:anti-DNA complexes in systemic lupus erythematosus. Detection and characterization by ultracentrifugation. J. Clin. Invest. 64, 191–198 (1979).
Morimoto, C., Sano, H., Abe, T., Homma, M. & Steinberg, A. D. Correlation between clinical activity of systemic lupus erythematosus and the amounts of DNA in DNA/anti-DNA antibody immune complexes. J. Immunol. 129, 1960–1965 (1982).
Tron, F., Letarte, J., Roque-Antunes Barreira, M. C. & Lesavre, P. Specific detection of circulating DNA:anti-DNA immune complexes in human systemic lupus erythematosus sera using murine monoclonal anti-DNA antibody. Clin. Exp. Immunol. 49, 481–487 (1982).
Bengtsson, A., Nezlin, R., Shoenfeld, Y. & Sturfelt, G. DNA levels in circulating immune complexes decrease at severe SLE flares-correlation with complement component C1q. J. Autoimmun. 13, 111–119 (1999).
Seredkina, N., Van Der Vlag, J., Berden, J., Mortensen, E. & Rekvig, O. P. Lupus nephritis: enigmas, conflicting models and an emerging concept. Mol. Med. 19, 161–169 (2013).
Krishnan, M. R., Wang, C. & Marion, T. N. Anti-DNA autoantibodies initiate experimental lupus nephritis by binding directly to the glomerular basement membrane in mice. Kidney Int. 82, 184–192 (2012).
Bruschi, M. et al. Glomerular autoimmune multicomponents of human lupus nephritis in vivo (2): planted antigens. J. Am. Soc. Nephrol. 26, 1905–1924 (2014).
Kalaaji, M., Mortensen, E., Jørgensen, L., Olsen, R. & Rekvig, O. P. Nephritogenic lupus antibodies recognize glomerular basement membrane-associated chromatin fragments released from apoptotic intraglomerular cells. Am. J. Pathol. 168, 1779–1792 (2006).
Mjelle, J. E., Kalaaji, M. & Rekvig, O. P. Exposure of chromatin and not high affinity for dsDNA determines the nephritogenic impact of anti-dsDNA antibodies in (NZBxNZW)F1 mice. Autoimmunity 42, 104–111 (2009).
Zykova, S. N., Tveita, A. A. & Rekvig, O. P. Renal DNase1 enzyme activity and protein expression is selectively shut down in murine and human membranoproliferative lupus nephritis. PLoS ONE 5, e12096 (2010).
Seredkina, N. & Rekvig, O. P. Acquired loss of renal nuclease activity is restricted to DNaseI and is an organ-selective feature in murine lupus nephritis. Am. J. Pathol. 179, 1120–1128 (2011).
Vallin, H., Perers, A., Alm, G. V. & Rönnblom, L. Anti-double-stranded DNA antibodies and immunostimulatory plasmid DNA in combination mimic the endogenous IFN-alpha inducer in systemic lupus erythematosus. J. Immunol. 163, 6306–6313 (1999).
Leadbetter, E. A. et al. Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416, 603–607 (2002).
Boulé, M. W. et al. Toll-like receptor 9-dependent and -independent dendritic cell activation by chromatin-immunoglobulin G complexes. J. Exp. Med. 199, 1631–1640 (2004).
Atianand, M. K. & Fitzgerald, K. A. Molecular basis of DNA recognition in the immune system. J. Immunol. 190, 1911–1918 (2013).
Bhat, N. & Fitzgerald, K. A. Recognition of cytosolic DNA by cGAS and other STING-dependent sensors. Eur. J. Immunol. 44, 634–640 (2014).
Hua, J., Kirou, K., Lee, C. & Crow, M. K. Functional assay of type I interferon in systemic lupus erythematosus plasma and association with anti-RNA binding protein autoantibodies. Arthritis Rheum. 54, 1906–1916 (2006).
Eloranta, M. L. et al. Regulation of the interferon-alpha production induced by RNA-containing immune complexes in plasmacytoid dendritic cells. Arthritis Rheum. 60, 2418–2427 (2009).
DeGiorgio, L. A. et al. A subset of lupus anti-DNA antibodies cross-reacts with the NR2 glutamate receptor in systemic lupus erythematosus. Nat. Med. 7, 1189–1193 (2001).
Kowal, C. et al. Human lupus autoantibodies against NMDA receptors mediate cognitive impairment. Proc. Natl Acad. Sci. USA 103, 19854–19859 (2006).
Mostoslavsky, G. et al. Lupus anti-DNA autoantibodies cross-react with a glomerular structural protein: a case for tissue injury by molecular mimicry. Eur. J. Immunol. 31, 1221–1227 (2001).
Zhao, Z. et al. Cross-reactivity of human lupus anti-DNA antibodies with alpha-actinin and nephritogenic potential. Arthritis Rheum. 52, 522–530 (2005).
Manson, J. J. et al. Relationship between anti-dsDNA, anti-nucleosome and anti-alpha-actinin antibodies and markers of renal disease in patients with lupus nephritis: a prospective longitudinal study. Arthritis Res. Ther. 11, R154 (2009).
Steiman, A. J. et al. Anti-dsDNA and antichromatin antibody isotypes in serologically active clinically quiescent systemic lupus erythematosus. J. Rheumatol. 42, 810–816 (2015).
Mannik, M., Merrill, C. E., Stamps, L. D. & Wener, M. H. Multiple autoantibodies form the glomerular immune deposits in patients with systemic lupus erythematosus. J. Rheumatol. 30, 1495–1504 (2003).
Xie, C., Liang, Z., Chang, S. & Mohan, C. Use of a novel elution regimen reveals the dominance of polyreactive antinuclear autoantibodies in lupus kidneys. Arthritis Rheum. 48, 2343–2352 (2003).
Leffler, J., Bengtsson, A. A. & Blom, A. M. The complement system in systemic lupus erythematosus: an update. Ann. Rheum. Dis. 73, 1601–1606 (2014).
Putterman, C. et al. Cell-bound complement activation products in systemic lupus erythematosus: comparison with anti-double-stranded DNA and standard complement measurements. Lupus Sci. Med. 1, e000056 (2014).
Nielsen, C. T., Østergaard, O., Johnsen, C., Jacobsen, S. & Heegaard, N. H. Distinct features of circulating microparticles and their relationship to clinical manifestations in systemic lupus erythematosus. Arthritis Rheum. 63, 3067–3077 (2011).
Nielsen, C. T. et al. Galectin-3 binding protein links circulating microparticles with electron dense glomerular deposits in lupus nephritis. Lupus 24, 1150–1160 (2015).
McCarty, G. A., Rice, J. R., Bembe, M. L. & Pisetsky, D. S. Independent expression of autoantibodies in systemic lupus erythematosus. J. Rheumatol. 9, 691–695 (1982).
Wahren, M. et al. Ro/SS-A and La/SS-B antibody level variation in patients with Sjögren's syndrome and systemic lupus erythematosus. J. Autoimmun. 11, 29–38 (1998).
Hassan, A. B., Lundberg, I. E., Isenberg, D. & Wahren-Herlenius, M. Serial analysis of Ro/SSA and La/SSB antibody levels and correlation with clinical disease activity in patients with systemic lupus erythematosus. Scand. J. Rheumatol. 31, 133–139 (2002).
Pisetsky, D. S., Grammer, A. C., Ning, T. C. & Lipsky, P. E. Are autoantibodies the targets of B-cell-directed therapy? Nat. Rev. Rheumatol. 7, 551–556 (2011).
Mathsson, L., Ahlin, E., Sjöwall, C., Skogh, T. & Rönnelid, J. Cytokine induction by circulating immune complexes and signs of in-vivo complement activation in systemic lupus erythematosus are associated with the occurrence of anti-Sjogren's syndrome A antibodies. Clin. Exp. Immunol. 147, 513–520 (2007).
Ahlin, E. et al. Autoantibodies associated with RNA are more enriched than anti-dsDNA antibodies in circulating immune complexes in SLE. Lupus 21, 586–595 (2012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Pisetsky, D. Anti-DNA antibodies — quintessential biomarkers of SLE. Nat Rev Rheumatol 12, 102–110 (2016). https://doi.org/10.1038/nrrheum.2015.151
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2015.151
This article is cited by
-
Affinity maturation generates pathogenic antibodies with dual reactivity to DNase1L3 and dsDNA in systemic lupus erythematosus
Nature Communications (2023)
-
Development of cyclopeptide inhibitors of cGAS targeting protein-DNA interaction and phase separation
Nature Communications (2023)
-
Risk of systemic lupus erythematosus flares according to autoantibody positivity at the time of diagnosis
Scientific Reports (2023)
-
Lupus nephritis or not? A simple and clinically friendly machine learning pipeline to help diagnosis of lupus nephritis
Inflammation Research (2023)
-
Brief report on the relation between complement C3a and anti dsDNA antibody in systemic lupus erythematosus
Scientific Reports (2022)