Key Points
-
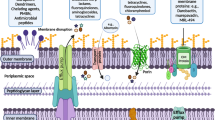
Porin modification is a major bacterial resistance strategy that restricts the influx of β-lactam and fluoroquinolone antibiotics.
-
Clinical multidrug resistant enterobacterial isolates that exhibit modified membrane permeability are highly prevalent.
-
Interactions between antibiotic molecules and porin channels govern translocation efficiency.
-
New physico-chemical techniques are being developed to assess drug–channel interactions and to quantify translocation through porins.
-
Computer modelling of the pathway of substrates through porins provides information on the orientation and interaction of substrates in the channel.
-
Quantification of antibiotic translocation provides new insights into how to optimize drug molecules so that they have sufficient permeation rates to circumvent multidrug resistance mechanisms.
Abstract
Gram-negative bacteria are responsible for a large proportion of antibiotic-resistant bacterial diseases. These bacteria have a complex cell envelope that comprises an outer membrane and an inner membrane that delimit the periplasm. The outer membrane contains various protein channels, called porins, which are involved in the influx of various compounds, including several classes of antibiotics. Bacterial adaptation to reduce influx through porins is an increasing problem worldwide that contributes, together with efflux systems, to the emergence and dissemination of antibiotic resistance. An exciting challenge is to decipher the genetic and molecular basis of membrane impermeability as a bacterial resistance mechanism. This Review outlines the bacterial response towards antibiotic stress on altered membrane permeability and discusses recent advances in molecular approaches that are improving our knowledge of the physico-chemical parameters that govern the translocation of antibiotics through porin channels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rice, L. B. Emerging issues in the management of infections caused by multidrug-resistant gram-negative bacteria. Cleve Clin. J. Med. 74, S12–S20 (2007).
Blot, S., Depuydt, P., Vandewoude, K. & De Bacquer, D. Measuring the impact of multidrug resistance in nosocomial infection. Curr. Opin. Infect. Dis. 20, 391–396 (2007).
Sheridan, C. Antiinfective biotechs face partnering gap. Nature Biotechnol. 23, 155–156 (2005).
Norrby, S. R., Nord, C. E. & Finch, R. Lack of development of new antimicrobial drugs: a potential serious threat to public health. Lancet Infect. Dis. 5, 115–119 (2005).
Li, X. Z. & Nikaido, H. Efflux-mediated drug resistance in bacteria. Drugs 64, 159–204 (2004).
Davin-Regli, A. et al. Membrane permeability and regulation of drug “influx and efflux” in enterobacterial pathogens. Curr. Drug Targets 9, 750–759 (2008).
Piddock, L. J. Multidrug-resistance efflux pumps — not just for resistance. Nature Rev. Microbiol. 4, 629–636 (2006).
Poole, K. Efflux-mediated antimicrobial resistance. J. Antimicrob. Chemother. 56, 20–51 (2005).
Nikaido, H. Molecular basis of bacterial outer membrane permeability revisited. Microbiol. Mol. Biol. Rev. 67, 593–656 (2003).
Delcour, A. H. Solute uptake through general porins. Front. Biosci. 8, D1055–D1071 (2003).
Schulz, G. E. The structure of bacterial outer membrane proteins. Biochim. Biophys. Acta 1565, 308–317 (2002).
Koebnik, R., Locher, K. P. & Van Gelder, P. Structure and function of bacterial outer membrane proteins: barrels in a nutshell. Mol. Microbiol. 37, 239–253 (2000).
Guillier, M., Gottesman, S. & Storz, G. Modulating the outer membrane with small RNAs. Genes Dev. 20, 2338–2348 (2006).
Pagès, J.-M. Role of bacterial porins in antibiotic susceptibility of Gram-negative bacteria in Bacterial and Eukaryotic Porins (ed. Benz, R.) 41–59 (Wiley-VCH, Weinheim,2004).
Davin-Régli, A. & Pagès, J.-M. in Antimicrobial Resistance in Bacteria (ed. Zmabiles-Cuevos, C. F.) 55–75 (Horizon Biosciences, Norfolk, 2006)
Bryskier, A. (ed.) Antimicrobial Agents: Antibacterials and Antifungals (ASM Press, Washington, 2005).
Kohanski, M. A., Dwyer, D. J., Hayete, B., Lawrence, C. A. & Collins, J. J. A common mechanism of cellular death induced by bactericidal antibiotics. Cell 130, 797–810 (2007).
Nestorovich, E. M., Sugawara, E., Nikaido, H. & Bezrukov, S. M. Pseudomonas aeruginosa porin OprF. Properties of the channel. J. Biol. Chem. 281, 16230–16237 (2006).
Sugawara, E., Nestorovich .E. M., Bezrukov, S. M. & Nikaido, H. Pseudomonas aeruginosa porin OprF exists in two different conformations. J. Biol. Chem. 281, 16220–16229 (2006).
Hancock, R. E. & Brinkman, F. S. Function of Pseudomonas porins in uptake and efflux. Annu. Rev. Microbiol. 56, 17–38 (2002).
Nikaido, H. Outer membrane barrier as a mechanism of antimicrobial resistance. Antimicrob. Agents Chemother. 33, 1831–1836 (1989).
Hasdemir, U. O., Chevalier, J., Nordmann, P. & Pagès, J.-M. Detection and prevalence of active drug efflux mechanism in various multidrug-resistant Klebsiella pneumoniae strains from Turkey. J. Clin. Microbiol. 42, 2701–2706 (2004).
Doménech-Sánchez, A. et al. Role of Klebsiella pneumoniae OmpK35 porin in antimicrobial resistance. Antimicrob. Agents Chemother. 47, 3332–3335 (2003).
Hernández-Allés, S. et al. Relationship between outer membrane alterations and susceptibility to antimicrobial agents in isogenic strains of Klebsiella pneumoniae. J. Antimicrob. Chemother. 46, 273–277 (2000).
Martínez-Martínez, L. et al. Energy-dependent accumulation of norfloxacin and porin expression in clinical isolates of Klebsiella pneumoniae and relationship to extended-spectrum β-lactamase production. Antimicrob. Agents Chemother. 46, 3926–3932 (2002).
Jacoby, G. A., Mills, D. M. & Chow, N. Role of β-lactamases and porins in resistance to ertapenem and other β-lactams in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 48, 3203–3206 (2004).
Kaczmarek, F. M., Dib-Hajj, F., Shang, W. & Gootz, T. D. High-level carbapenem resistance in a Klebsiella pneumoniae clinical isolate is due to the combination of bla(ACT-1) β-lactamase production, porin OmpK35/36 insertional inactivation, and down-regulation of the phosphate transport porin phoe. Antimicrob. Agents Chemother. 50, 3396–3406 (2006).
Mena, A. et al. Characterization of a large outbreak by CTX-M-1-producing Klebsiella pneumoniae and mechanisms leading to in vivo carbapenem resistance development. J. Clin. Microbiol. 44, 2831–2837 (2006).
Loli, A. et al. Sources of diversity of carbapenem resistance levels in Klebsiella pneumoniae carrying blaVIM-1. J. Antimicrob. Chemother. 58, 669–672 (2006).
Elliott, E. et al. In vivo development of ertapenem resistance in a patient with pneumonia caused by Klebsiella pneumoniae with an extended-spectrum β-lactamase. Clin. Infect. Dis. 42, e95–e98 (2006).
Medeiros, A. A., O'Brien, T. F., Rosenberg, E. Y. & Nikaido, H. Loss of OmpC porin in a strain of Salmonella typhimurium causes increased resistance to cephalosporins during therapy. J. Infect. Dis. 156, 751–757 (1987).
Yoshimura, F. & Nikaido, H. Diffusion of β-lactam antibiotics through the porin channels of Escherichia coli K-12. Antimicrob. Agents Chemother. 27, 84–92 (1985). The first complete study on the diffusion rate of β-lactam molecules through E. coli porins.
Ferenci, T. Maintaining a healthy SPANC balance through regulatory and mutational adaptation. Mol. Microbiol. 57, 1–8 (2005).
Prilipov, A., Phale, P. S., Koebnik, R., Widmer, C. & Rosenbusch, J. P. Identification and characterization of two quiescent porin genes, nmpC and ompN, in Escherichia coli BE. J. Bacteriol. 180, 3388–3392 (1998).
Doménech-Sánchez, A., Hernández-Allés, S., Martínez-Martínez, L., Benedí, V. J. & Albertí, S. Identification and characterization of a new porin gene of Klebsiella pneumoniae: its role in β-lactam antibiotic resistance. J. Bacteriol. 181, 2726–2732 (1999).
Bornet, C., Davin-Régli, A., Bosi, C., Pagès, J.-M. & Bollet, C. Imipenem resistance of Enterobacter aerogenes mediated by outer membrane permeability. J. Clin. Microbiol. 38, 1048–1052 (2000).
Bosi, C. et al. Most Enterobacter aerogenes strains in France belong to a prevalent clone. Clin. Microbiol. 37, 2165–2169 (1999).
Bornet, C. et al. Imipenem and expression of multidrug efflux pump in Enterobacter aerogenes. Biochem. Biophys. Res. Commun. 301, 985–990 (2003).
Charrel, R. N., Pagès, J.-M., De Micco, P. & Malléa, M. Prevalence of outer membrane porin alteration in β-lactam-antibiotic-resistant Enterobacter aerogenes. Antimicrob. Agents Chemother. 40, 2854–2858 (1996).
Thiolas, A., Bollet, C., La Scola, B., Raoult, D. & Pagès, J.-M. Successive emergence of Enterobacter aerogenes strains resistant to imipenem and colistin in a patient. Antimicrob. Agents Chemother. 49, 1354–1358 (2005).
Szabó, D. et al. Outer membrane protein changes and efflux pump expression together may confer resistance to ertapenem in Enterobacter cloacae. Antimicrob. Agents Chemother. 50, 2833–2835 (2006).
Randall, L. P. & Woodward, M. J. The multiple antibiotic resistance (mar) locus and its significance. Res. Vet. Sci. 72, 87–93 (2002).
Hernández-Allés, S. et al. Development of resistance during antimicrobial therapy caused by insertion sequence interruption of porin genes. Antimicrob. Agents Chemother. 43, 937–939 (1999).
Mena, A. et al. Characterization of a large outbreak by CTX-M-1-producing Klebsiella pneumoniae and mechanisms leading to in vivo carbapenem resistance development. J. Clin. Microbiol. 44, 2831–2837 (2006).
Cai, J. C., Zhou, H. W., Zhang, R. & Chen, G. X. Emergence of Serratia marcescens, Klebsiella pneumoniae, and Escherichia coli isolates possessing the plasmid-mediated carbapenem-hydrolyzing β-lactamase KPC-2 in intensive care units of a Chinese hospital. Antimicrob. Agents Chemother. 52, 2014–2018 (2008).
Simonet, V., Malléa, M., Fourel, D., Bolla, J. M. & Pagès, J.-M. Crucial domains are conserved in Enterobacteriaceae porins. FEMS Microbiol. Lett. 136, 91–97 (1996).
Low, A. S., MacKenzie, F. M., Gould, I. M. & Booth, I. R. Protected environments allow parallel evolution of a bacterial pathogen in a patient subjected to long-term antibiotic therapy. Mol. Microbiol. 42, 619–630 (2001).
Malléa, M. et al. Porin alteration and active efflux: two in vivo drug resistance strategies used by Enterobacter aerogenes. Microbiology 144, 3003–3009 (1998).
Dé, E. et al. A new mechanism of antibiotic resistance in Enterobacteriaceae induced by a structural modification of the major porin. Mol. Microbiol. 41, 189–198 (2001). The first description of a clinical isolate with a mutation in internal loop 3.
Jeanteur, D. et al. Structural and functional alterations of a colicin-resistant mutant of OmpF porin from Escherichia coli. Proc. Natl Acad. Sci. USA 91, 10675–10679 (1994).
Simonet, V., Malléa, M. & Pagès, J.-M. Substitutions in the eyelet region disrupt cefepime diffusion through the Escherichia coli OmpF channel. Antimicrob. Agents Chemother. 44, 311–315 (2000).
Thiolas, A., Bornet, C., Davin-Régli, A., Pagès, J.-M. & Bollet, C. Resistance to imipenem, cefepime, and cefpirome associated with mutation in Omp36 osmoporin of Enterobacter aerogenes. Biochem. Biophys. Res. Commun. 317, 851–856 (2004).
Chevalier, J., Pagès, J.-M. & Malléa, M. In vivo modification of porin activity conferring antibiotic resistance to Enterobacter aerogenes. Biochem. Biophys. Res. Commun. 266, 248–251 (1999).
Olesky, M., Hobbs, M. & Nicholas, R. A. Identification and analysis of amino acid mutations in porin IB that mediate intermediate-level resistance to penicillin and tetracycline in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 46, 2811–2820 (2002).
Olesky, M., Zhao, S., Rosenberg, R. L. & Nicholas, R. A. Porin-mediated antibiotic resistance in Neisseria gonorrhoeae: ion, solute, and antibiotic permeation through PIB proteins with penB mutations. J. Bacteriol. 188, 2300–2308 (2006).
Veal, W. L., Nicholas, R. A. & Shafer, W. M. Overexpression of the MtrC–MtrD–MtrE efflux pump due to an MtrR mutation is required for chromosomally mediated penicillin resistance in Neisseria gonorrhoeae. J. Bacteriol. 184, 5619–5624 (2002).
Shafer, W. M. & Folster, J. P. Towards an understanding of chromosomally mediated penicillin resistance in Neisseria gonorrhoeae: evidence for a porin-efflux pump collaboration. J. Bacteriol. 188, 2297–2299 (2006).
Ochs, M. M., Bains, M. & Hancock, R. E. Role of putative loops 2 and 3 in imipenem passage through the specific porin OprD of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 44, 1983–1985 (2000).
Wolter, D. J., Hanson, N. D. & Lister, P. D. Insertional inactivation of oprD in clinical isolates of Pseudomonas aeruginosa leading to carbapenem resistance. FEMS Microbiol. Lett. 236, 137–143 (2004).
Epp, S. F. et al. C-terminal region of Pseudomonas aeruginosa outer membrane porin OprD modulates susceptibility to meropenem. Antimicrob. Agents Chemother. 45, 1780–1787 (2001).
Biswas, S., Mohammad, M. M., Patel, D. R., Movileanu, L. & van den Berg, B. Structural insight into OprD substrate specificity. Nature Struct. Mol. Biol. 14, 1108–1109 (2007).
El Amin, N. et al. Carbapenem resistance mechanisms in Pseudomonas aeruginosa: alterations of porin OprD and efflux proteins do not fully explain resistance patterns observed in clinical isolates. APMIS 113, 187–196 (2005).
Luckey, M. & Nikaido, H. Specificity of diffusion channels produced by λ phage receptor protein of Escherichia coli. Proc. Natl Acad. Sci. USA 77, 167–171 (1980).
Nikaido, H. & Rosenberg, E. Y. Porin channels in Escherichia coli: studies with liposomes reconstituted from purified proteins. J. Bacteriol. 153, 241–252 (1983).
Rodrigues, C., Gameiro, P., Prieto, M. & de Castro, B. Interaction of rifampicin and isoniazid with large unilamellar liposomes: spectroscopic location studies. Biochimica Biophysica Acta 1620, 151–159 (2003).
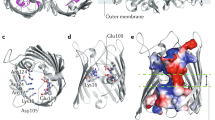
Cowan, S. W. et al. Crystal structures explain functional properties of two E. coli porins. Nature 358, 727–733 (1992). The three-dimensional structures of OmpF and PhoE explained the channel properties of E. coli porins.
Baslé, A., Rummel, G., Storici, P., Rosenbusch, J. P. & Schirmer, T. Crystal structure of osmoporin OmpC from E. coli at 2A. J. Mol. Biol. 362, 933–942 (2006). Reported the three-dimensional structure of E. coli OmpC porin.
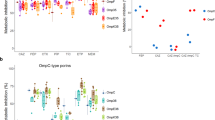
Danelon, C., Nestrovich, E. M., Winterhalter, M., Ceccarelli, M. & Bezrukov, S. M. Interaction of zwitterionic penicillins with the OmpF channel facilitates their translocation. Biophys. J. 90, 1617–1627 (2006).
Nestorovich, E. M., Danelon, C., Winterhalter, M. & Bezrukov, S. M. Designed to penetrate: time-resolved interaction of single antibiotic molecules with bacterial pores. Proc. Natl Acad. Sci. USA 99, 9789–9794 (2002). The first characterization of the β-lactam permeation through OmpF on a single molecular level.
Danelon, C., Brando, T. & Winterhalter, M. Probing the orientation of reconstituted maltoporin channels at the single-protein level. J. Biol. Chem. 278, 35542–35551 (2003).
Kullman, L, Winterhalter, M. & Bezrukov, S. M. Transport of maltodextrins through maltoporin. A single-channel study. Biophys. J. 82, 803–812 (2002).
Bezrukov, S. M. & Winterhalter, M. Examining noise sources at the single-molecule level: 1/f noise of an open maltoporin channel. Phys. Rev. Lett. 85, 202–205 (2000).
Nekolla, S., Andersen, C. & Benz, R. Noise analysis of ion current through the open and the sugar-induced closed state of the LamB channel of Escherichia coli outer membrane: evaluation of the sugar binding kinetics to the channel interior Biophys. J. 66, 1388–1397 (1994).
Schwarz, G., Danelon, C. & Winterhalter, M. On translocation through a membrane channel via an internal binding site: kinetics and voltage dependence. Biophy. J. 84, 2990–2998 (2003).
Vidal, S., Bredin, J., Pagès, J.-M. & Barbe, J. β-lactam screening by specific residues of the OmpF eyelet. J. Med. Chem. 48, 1395–1400 (2005).
Berezhkovskii, A. M. & Bezrukov, S. M. Optimizing transport of metabolites through large channels: molecular sieves with and without binding. Biophys. J. 88, L17–L19 (2005).
Bauer, W. R. & Nadler, W. Molecular transport through channels and pores: effects of in-channel interactions and blocking. Proc. Natl Acad. Sci. USA 103, 11446–11451 (2006).
Bezrukov, S. M., Berezhkovskii, A. M. & Szabo, A. Diffusion model of solute dynamics in a membrane channel: mapping onto the two-site model and optimizing the flux. J. Chem. Phys. 127, 115101 (2007).
Ceccarelli, M., Danelon, C., Laio, A. & Parrinello, M. Microscopic mechanism of antibiotics translocation through a porin. Biophys. J. 87, 58–64 (2004).
Giuliodori, A. M., Gualerzi, C. O., Soto, S., Vila, J. & Tavío, M. M. Review on bacterial stress topics. Ann. NY Acad. Sci. 1113, 95–104 (2007).
Valentin-Hansen, P., Johansen, J. & Rasmussen, A. A. Small RNAs controlling outer membrane porins. Curr. Opin. Microbiol. 10, 152–155 (2007).
Viveiros, M. et al. Antibiotic stress, genetic response and altered permeability of E. coli. PLoS ONE 2, e365 (2007).
Baslé, A. & Delcour, A. H. Effect of two polyamine toxins on the bacterial porin OmpF. Biochem. Biophys. Res. Commun. 285, 550–554 (2001).
Iyer, R., Wu, Z., Woster, P. M. & Delcour, A. H. Molecular basis for the polyamine-ompF porin interactions: inhibitor and mutant studies. J. Mol. Biol. 297, 933–945 (2000).
Iyer, R. & Delcour, A. H. Complex inhibition of OmpF and OmpC bacterial porins by polyamines. J. Biol. Chem. 272, 18595–18601 (1997).
Vidal, S. et al. Computer simulation of spermine–porin channel interactions. In Vivo 16, 111–116 (2002).
Bredin, J., Simonet, V., Iyer, R., Delcour, A. H. & Pagès, J.-M. Colicins, spermine and cephalosporins: a competitive interaction with the OmpF eyelet. Biochem. J. 376, 245–252 (2003).
Chevalier, J., Malléa, M. & Pagès, J.-M. Comparative aspects of the diffusion of norfloxacin, cefepime and spermine through the F porin channel of Enterobacter cloacae. Biochem. J. 348, 223–227 (2000).
Samartzidou, H. & Delcour, A. H. Excretion of endogenous cadaverine leads to a decrease in porin-mediated outer membrane permeability. J. Bacteriol. 181, 791–798 (1999).
Samartzidou, H., Mehrazin, M., Xu, Z., Benedik, M. J. & Delcour, A. H. Cadaverine inhibition of porin plays a role in cell survival at acidic pH. J. Bacteriol. 185, 13–19 (2003).
Bower, J. M. & Mulvey, M. A. Polyamine-mediated resistance of uropathogenic Escherichia coli to nitrosative stress. J. Bacteriol. 188, 928–933 (2006).
Dutzler, R. et al. Crystal structure and functional characterization of OmpK36, the osmoporin of Klebsiella pneumoniae. Structure 7, 425–434 (1999).
Mach, T., Chimerel, C., Fritz, J., Fertig, N., Winterhalter, M. & Fütterer, C. Miniaturized planar lipid bilayer: increased stability, low electric noise and fast fluid perfusion. Analytical Bionanalyt. Chem. 390, 341–346 (2008).
Acknowledgements
Support for this project was obtained through European Union grant MRTN-CT-2005–019335 “Translocation”, COST Action BM0701 “ATENS” and from the Université de la Méditerranée and Service de Santé des Armées.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Nosocomial
-
Hospital-acquired infection.
- Conductance
-
A measure of translocated charges per unit time and voltage gradient.
- Selectivity
-
The translocation efficiency of a channel for a particular type of ion with respect to another ion.
- Voltage gating
-
Effect observed for some channels whereby a high voltage gradient causes a sudden closure of the ion current. The molecular origins remain unsolved.
- β-lactam
-
A major family of antibiotic molecules.
- Antibiotherapy
-
A therapy that uses antibiotics to treat infections.
- Cephalosporins and carbapenems
-
Two subclasses of the β-lactam family.
- Bacteraemia
-
A medical condition in which bacteria enter the bloodstream.
- Permeation
-
Diffusion through a membrane, either through the lipid or through channels or carriers.
- Metadynamics modelling
-
A method to simulate rare events on the basis of the choice of the relevant variables of a process and on the acceleration of these variables.
Rights and permissions
About this article
Cite this article
Pagès, JM., James, C. & Winterhalter, M. The porin and the permeating antibiotic: a selective diffusion barrier in Gram-negative bacteria. Nat Rev Microbiol 6, 893–903 (2008). https://doi.org/10.1038/nrmicro1994
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro1994
This article is cited by
-
Cephalosporin resistance, tolerance, and approaches to improve their activities
The Journal of Antibiotics (2024)
-
Exploiting the synergistic antibacterial activity of shikimic acid and ceftiofur against methicillin-resistant Staphylococcus aureus
World Journal of Microbiology and Biotechnology (2024)
-
Molecular mechanism of engineered Zymomonas mobilis to furfural and acetic acid stress
Microbial Cell Factories (2023)
-
Molecular characterization and in-depth genomic analysis to unravel the pathogenic features of an environmental isolate Enterobacter sp. S-33
International Microbiology (2023)
-
Porin-Mediated Carbapenem Resistance in Klebsiella pneumoniae: an Alarming Threat to Global Health
Current Clinical Microbiology Reports (2023)