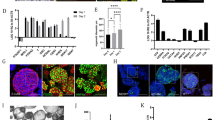
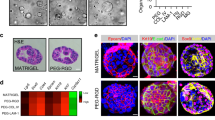
Abstract
Adult somatic tissues have proven difficult to expand in vitro, largely because of the complexity of recreating appropriate environmental signals in culture. We have overcome this problem recently and developed culture conditions for adult stem cells that allow the long-term expansion of adult primary tissues from small intestine, stomach, liver and pancreas into self-assembling 3D structures that we have termed 'organoids'. We provide a detailed protocol that describes how to grow adult mouse and human liver and pancreas organoids, from cell isolation and long-term expansion to genetic manipulation in vitro. Liver and pancreas cells grow in a gel-based extracellular matrix (ECM) and a defined medium. The cells can self-organize into organoids that self-renew in vitro while retaining their tissue-of-origin commitment, genetic stability and potential to differentiate into functional cells in vitro (hepatocytes) and in vivo (hepatocytes and endocrine cells). Genetic modification of these organoids opens up avenues for the manipulation of adult stem cells in vitro, which could facilitate the study of human biology and allow gene correction for regenerative medicine purposes. The complete protocol takes 1-4 weeks to generate self-renewing 3D organoids and to perform genetic manipulation experiments. Personnel with basic scientific training can conduct this protocol.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Huch, M. & Koo, B.K. Modeling mouse and human development using organoid cultures. Development 142, 3113–3125 (2015).
Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011).
Lancaster, M.A. et al. Cerebral organoids model human brain development and microcephaly. Nature 501, 373–379 (2013).
McCracken, K.W., Howell, J.C., Wells, J.M. & Spence, J.R. Generating human intestinal tissue from pluripotent stem cells. Nat. Protoc. 6, 1920–1928 (2011).
McCracken, K.W. et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature 516, 400–404 (2014).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Barker, N. et al. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units. Cell Stem Cell 6, 25–36 (2010).
Huch, M. et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature 494, 247–250 (2013).
Huch, M. et al. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell 160, 299–312 (2015).
Huch, M. et al. Unlimited in vitro expansion of adult bi-potent pancreas progenitors through the Lgr5/R-spondin axis. EMBO J. 32, 2708–2721 (2013).
Boj, S.F. et al. Organoid models of human and mouse ductal pancreatic cancer. Cell 160, 324–338 (2015).
Koo, B.K. et al. Controlled gene expression in primary Lgr5 organoid cultures. Nat. Methods 9, 81–83 (2012).
Schwank, G. et al. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13, 653–658 (2013).
Zaret, K.S. Genetic programming of liver and pancreas progenitors: lessons for stem-cell differentiation. Nat. Rev. Genet. 9, 329–340 (2008).
Duncan, A.W., Dorrell, C. & Grompe, M. Stem cells and liver regeneration. Gastroenterology 137, 466–481 (2009).
Zhao, R. & Duncan, S.A. Embryonic development of the liver. Hepatology 41, 956–967 (2005).
Zorn, A.M. Liver development. In StemBook 31 Oct 2008 (ed. Harvard Stem Cell Institute, Cambridge, MA, 2008).
Shih, H.P., Wang, A. & Sander, M. Pancreas organogenesis: from lineage determination to morphogenesis. Annu. Rev. Cell Dev. Biol. 29, 81–105 (2013).
Michalopoulos, G.K. et al. Histological organization in hepatocyte organoid cultures. Am. J. Pathol. 159, 1877–1887 (2001).
Greggio, C. et al. Artificial three-dimensional niches deconstruct pancreas development. Development 140, 4452–4462 (2013).
D'Amour, K.A. et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 24, 1392–1401 (2006).
Pagliuca, F.W. et al. Generation of functional human pancreatic β cells. Cell 159, 428–439 (2014).
Zaret, K.S. & Grompe, M. Generation and regeneration of cells of the liver and pancreas. Science 322, 1490–1494 (2008).
Bort, R., Signore, M., Tremblay, K., Martinez Barbera, J.P. & Zaret, K.S. Hex homeobox gene controls the transition of the endoderm to a pseudostratified, cell emergent epithelium for liver bud development. Dev. Biol. 290, 44–56 (2006).
Francis, H. et al. cAMP stimulates the secretory and proliferative capacity of the rat intrahepatic biliary epithelium through changes in the PKA/Src/MEK/ERK1/2 pathway. J. Hepatol. 41, 528–537 (2004).
McCright, B., Lozier, J. & Gridley, T. A mouse model of Alagille syndrome: Notch2 as a genetic modifier of Jag1 haploinsufficiency. Development 129, 1075–1082 (2002).
Ahlgren, U., Jonsson, J. & Edlund, H. The morphogenesis of the pancreatic mesenchyme is uncoupled from that of the pancreatic epithelium in IPF1/PDX1-deficient mice. Development 122, 1409–1416 (1996).
Donehower, L.A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992).
Baker, S.J. et al. Chromosome 17 deletions and p53 gene mutations in colorectal carcinomas. Science 244, 217–221 (1989).
Ingalls, A.M., Dickie, M.M. & Snell, G.D. Obese, a new mutation in the house mouse. J. Hered. 41, 317–318 (1950).
Evers, B. & Jonkers, J. Mouse models of BRCA1 and BRCA2 deficiency: past lessons, current understanding and future prospects. Oncogene 25, 5885–5897 (2006).
Andersson-Rolf, A., Fink, J., Mustata, R.C. & Koo, B.K. A video protocol of retroviral infection in primary intestinal organoid culture. J. Vis. Exp. 90 e51765 (2014).
Koo, B.K. et al. Tumour suppressor RNF43 is a stem-cell E3 ligase that induces endocytosis of Wnt receptors. Nature 488, 665–669 (2012).
Davis, H.E., Morgan, J.R. & Yarmush, M.L. Polybrene increases retrovirus gene transfer efficiency by enhancing receptor-independent virus adsorption on target cell membranes. Biophys. Chem. 97, 159–172 (2002).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013).
Van Lidth de Jeude, J.F., Vermeulen, J.L., Montenegro-Miranda, P.S., Van den Brink, G.R. & Heijmans, J. A protocol for lentiviral transduction and downstream analysis of intestinal organoids. J. Vis. Exp. 98, e52531 (2015).
Wang, N. et al. Adenovirus-mediated efficient gene transfer into cultured three-dimensional organoids. PLoS One 9, e93608 (2014).
Klaunig, J.E. et al. Mouse liver cell culture. I. Hepatocyte isolation. In Vitro 17, 913–925 (1981).
Korinek, V. et al. Constitutive transcriptional activation by a beta-catenin-Tcf complex in APC−/− colon carcinoma. Science 275, 1784–1787 (1997).
de Lau, W. et al. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature. 476, 293–297 (2011).
Acknowledgements
L.B. is supported by an EMBO postdoctoral fellowship (EMBO ALTF 794-2014). C.J.H. is supported by a Cambridge Stem Cell Institute Seed Fund award and the Herchel Smith Fund. B.-K.K. is supported by a Sir Henry Dale Fellowship from the Wellcome Trust and the Royal Society. M.H. is a Wellcome Trust Sir Henry Dale Fellow and is jointly funded by the Wellcome Trust and the Royal Society (104151/Z/14/Z). A.A.-R. is funded by an MRC PhD fellowship. We gratefully acknowledge the kind gift of 293T-HA-Rspo1-Fc cells from Calvin Kuo, Stanford University.
Author information
Authors and Affiliations
Contributions
M.H. and S.F.B. developed the culture system for liver and pancreas. B.-K.K. developed the genetic manipulation of organoid cultures. H.C. supervised the development of organoid cultures and their genetic manipulation. L.B., A.A.-R. and C.J.H. prepared figures. L.B., A.A.-R., C.J.H., S.F.B. and M.H. wrote the manuscript. All authors commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1 and 2 (PDF 565 kb)
Rights and permissions
About this article
Cite this article
Broutier, L., Andersson-Rolf, A., Hindley, C. et al. Culture and establishment of self-renewing human and mouse adult liver and pancreas 3D organoids and their genetic manipulation. Nat Protoc 11, 1724–1743 (2016). https://doi.org/10.1038/nprot.2016.097
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.097
This article is cited by
-
LUBAC-mediated M1 Ub regulates necroptosis by segregating the cellular distribution of active MLKL
Cell Death & Disease (2024)
-
Liver organoid culture methods
Cell & Bioscience (2023)
-
Growth differentiation factor 7 autocrine signaling promotes hepatic progenitor cell expansion in liver fibrosis
Stem Cell Research & Therapy (2023)
-
Gene expression in organoids: an expanding horizon
Biology Direct (2023)
-
Towards a better understanding of diabetes mellitus using organoid models
Nature Reviews Endocrinology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.