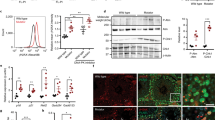
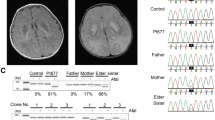
Abstract
Mitochondrial DNA (mtDNA) depletion syndrome (MDS; MIM 251880) is a prevalent cause of oxidative phosphorylation disorders characterized by a reduction in mtDNA copy number. The hitherto recognized disease mechanisms alter either mtDNA replication (POLG (ref. 1)) or the salvage pathway of mitochondrial deoxyribonucleosides 5′-triphosphates (dNTPs) for mtDNA synthesis (DGUOK (ref. 2), TK2 (ref. 3) and SUCLA2 (ref. 4)). A last gene, MPV17 (ref. 5), has no known function. Yet the majority of cases remain unexplained. Studying seven cases of profound mtDNA depletion (1–2% residual mtDNA in muscle) in four unrelated families, we have found nonsense, missense and splice-site mutations and in-frame deletions of the RRM2B gene, encoding the cytosolic p53-inducible ribonucleotide reductase small subunit. Accordingly, severe mtDNA depletion was found in various tissues of the Rrm2b−/− mouse. The mtDNA depletion triggered by p53R2 alterations in both human and mouse implies that p53R2 has a crucial role in dNTP supply for mtDNA synthesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ferrari, G. et al. Infantile hepatocerebral syndromes associated with mutations in the mitochondrial DNA polymerase-γA. Brain 128, 723–731 (2005).
Mandel, H. et al. The deoxyguanosine kinase gene is mutated in individuals with depleted hepatocerebral mitochondrial DNA. Nat. Genet. 29, 337–341 (2001).
Saada, A. et al. Mutant mitochondrial thymidine kinase in mitochondrial DNA depletion myopathy. Nat. Genet. 29, 342–344 (2001).
Elpeleg, O. et al. Deficiency of the ADP-forming succinyl-CoA synthase activity is associated with encephalomyopathy and mitochondrial DNA depletion. Am. J. Hum. Genet. 76, 1081–1086 (2005).
Spinazzola, A. et al. MPV17 encodes an inner mitochondrial membrane protein and is mutated in infantile hepatic mitochondrial DNA depletion. Nat. Genet. 38, 570–575 (2006).
Nordlund, P. & Reichard, P. Ribonucleotide reductases. Annu. Rev. Biochem. 75, 681–706 (2006).
Tanaka, H. et al. A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature 404, 42–49 (2000).
Kimura, T. et al. Impaired function of p53R2 in Rrm2b-null mice causes severe renal failure through attenuation of dNTP pools. Nat. Genet. 34, 440–445 (2003).
Comeau, S.R., Gatchell, D.W., Vajda, S. & Camacho, C.J. ClusPro: a fully automated algorithm for protein-protein docking. Nucleic Acids Res. 32, W96–W99 (2004).
Comeau, S.R., Gatchell, D.W., Vajda, S. & Camacho, C.J. ClusPro: an automated docking and discrimination method for the prediction of protein complexes. Bioinformatics 20, 45–50 (2004).
Eklund, H., Uhlin, U., Farnegardh, M., Logan, D.T. & Nordlund, P. Structure and function of the radical enzyme ribonucleotide reductase. Prog. Biophys. Mol. Biol. 77, 177–268 (2001).
Lycksell, P.O., Ingemarson, R., Davis, R., Graslund, A. & Thelander, L. 1H NMR studies of mouse ribonucleotide reductase: the R2 protein carboxyl-terminal tail, essential for subunit interaction, is highly flexible but becomes rigid in the presence of protein R1. Biochemistry 33, 2838–2842 (1994).
Kolberg, M., Strand, K.R., Graff, P. & Andersson, K.K. Structure, function, and mechanism of ribonucleotide reductases. Biochim. Biophys. Acta 1699, 1–34 (2004).
Hakansson, P., Hofer, A. & Thelander, L. Regulation of mammalian ribonucleotide reduction and dNTP pools after DNA damage and in resting cells. J. Biol. Chem. 281, 7834–7841 (2006).
Chabes, A.L., Pfleger, C.M., Kirschner, M.W. & Thelander, L. Mouse ribonucleotide reductase R2 protein: a new target for anaphase-promoting complex-Cdh1-mediated proteolysis. Proc. Natl. Acad. Sci. USA 100, 3925–3929 (2003).
Zhou, B. et al. The human ribonucleotide reductase subunit hRRM2 complements p53R2 in response to UV-induced DNA repair in cells with mutant p53. Cancer Res. 63, 6583–6594 (2003).
Matoba, S. et al. p53 regulates mitochondrial respiration. Science 312, 1650–1653 (2006).
Liu, Z. & Butow, R.A. Mitochondrial retrograde signaling. Annu. Rev. Genet. 40, 159–185 (2006).
Butow, R.A. & Avadhani, N.G. Mitochondrial signaling: the retrograde response. Mol. Cell 14, 1–15 (2004).
Paquis-Flucklinger, V. et al. Early-onset fatal encephalomyopathy associated with severe mtDNA depletion. Eur. J. Pediatr. 154, 557–562 (1995).
Abecasis, G.R., Cherny, S.S., Cookson, W.O. & Cardon, L.R. Merlin—rapid analysis of dense genetic maps using sparse gene flow trees. Nat. Genet. 30, 97–101 (2002).
Rustin, P. et al. Biochemical and molecular investigations in respiratory chain deficiencies. Clin. Chim. Acta 228, 35–51 (1994).
Sarzi, E. et al. Mitochondrial DNA depletion is a prevalent cause of multiple respiratory chain deficiency in childhood. J. Pediatr. (in the press).
Schwede, T., Kopp, J., Guex, N. & Peitsch, M.C. SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res. 31, 3381–3385 (2003).
Hooft, R.W., Vriend, G., Sander, C. & Abola, E.E. Errors in protein structures. Nature 381, 272 (1996).
Guex, N. & Peitsch, M.C. SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis 18, 2714–2723 (1997).
Acknowledgements
We thank T. Soussi for discussion and J. Bentata, B. Chabrol, C. Fallet-Bianco and J.M. Liet for referring the study subjects. This research was supported in part by the Association Française contre les Myopathies, Mitocircle European Union Project (contract number LSHB-CT-2004-005260), Eumitocombat within the European Union Sixth Framework Program for Research, Priority 1 'Life sciences, genomics and biotechnology for health' (contract number LSHM-CT-2004-503116) and the French Agence Nationale pour la Recherche.
Author information
Authors and Affiliations
Contributions
A.B. identified the p53R2 mutations and performed the protein blot and mtDNA quantification in mouse, L.M. performed protein blot analysis, V.S. performed the modeling of ribonucleotide reductase, J.-P.J. performed the linkage analysis using Merlin software, E.S. performed the mitochondrial DNA quantification, S.A. was in charge of cell culture, D.C. identified the respiratory chain enzyme activity, P.d.L. was responsible for clinical management of subjects 3, 4 and 7 and their diagnosis, V.P.-F. was responsible for clinical management of subject 7 and her diagnosis, H.A. and Y.N. produced the Rrm2b−/− mouse, A.M. was responsible for clinical management of subjects 1 and 2 and wrote the paper, and A.R. designed the project and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Sequence analysis of the RRM2B gene. (PDF 17 kb)
Supplementary Fig. 2
Sequence alignment of p53R2 and R2 proteins from human and nonhuman sources. (PDF 6 kb)
Supplementary Fig. 3
Protein blot analysis of Bax and Bcl2 in subject and control cells. (PDF 12 kb)
Supplementary Table 1
Oligonucleotide primers. (PDF 3 kb)
Rights and permissions
About this article
Cite this article
Bourdon, A., Minai, L., Serre, V. et al. Mutation of RRM2B, encoding p53-controlled ribonucleotide reductase (p53R2), causes severe mitochondrial DNA depletion. Nat Genet 39, 776–780 (2007). https://doi.org/10.1038/ng2040
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng2040
This article is cited by
-
Identification of two novel RRM2B variants associated with autosomal recessive progressive external ophthalmoplegia in a family with pseudodominant inheritance pattern
Journal of Human Genetics (2023)
-
Impact of N221S missense mutation in human ribonucleotide reductase small subunit b on mitochondrial DNA depletion syndrome
Scientific Reports (2023)
-
Ribonucleotide reductase M2B in the myofibers modulates stem cell fate in skeletal muscle
npj Regenerative Medicine (2022)
-
DTYMK is essential for genome integrity and neuronal survival
Acta Neuropathologica (2022)
-
The regulatory roles of p53 in cardiovascular health and disease
Cellular and Molecular Life Sciences (2021)