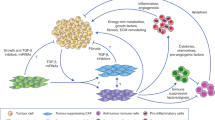
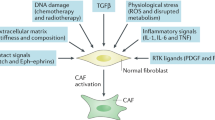
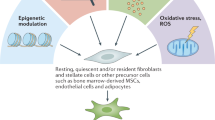
Key Points
-
Resting or quiescent adult fibroblasts are indolent and probably remnants of mesenchymal cells during organ development.
-
Resting fibroblasts serve as precursors of activated fibroblasts including myofibroblasts.
-
Resting fibroblasts share some features with adult tissue stem cells and embryonic stem cells.
-
Activated fibroblasts can differentiate into adipocytes and chondrocytes and exhibit the potential to be programmed into induced pluripotent stem cells, in part because of their epigenetic and transcriptomic state, which favours their reprogramming efficiency.
-
Resting fibroblasts can differentiate into active fibroblasts that are synthetically active and can generate growth factors and extracellular matrix.
-
Cancer metabolism is influenced by activated fibroblasts.
-
Activated fibroblasts recruit immune cells and regulate tumour immunity.
-
Activated fibroblasts modulate chemoresistance.
-
Angiogenesis can be stimulated by activated fibroblasts.
-
It is now generally accepted that cancer-associated fibroblasts are a heterogeneous population with distinct functions.
-
Cancer-associated fibroblasts can serve as positive and negative regulators of tumour progression.
Abstract
Among all cells, fibroblasts could be considered the cockroaches of the human body. They survive severe stress that is usually lethal to all other cells, and they are the only normal cell type that can be live-cultured from post-mortem and decaying tissue. Their resilient adaptation may reside in their intrinsic survival programmes and cellular plasticity. Cancer is associated with fibroblasts at all stages of disease progression, including metastasis, and they are a considerable component of the general host response to tissue damage caused by cancer cells. Cancer-associated fibroblasts (CAFs) become synthetic machines that produce many different tumour components. CAFs have a role in creating extracellular matrix (ECM) structure and metabolic and immune reprogramming of the tumour microenvironment with an impact on adaptive resistance to chemotherapy. The pleiotropic actions of CAFs on tumour cells are probably reflective of them being a heterogeneous and plastic population with context-dependent influence on cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011). A comprehensive review of cancer.
Ronnov-Jessen, L., Petersen, O. W. & Bissell, M. J. Cellular changes involved in conversion of normal to malignant breast: importance of the stromal reaction. Physiol. Rev. 76, 69–125 (1996).
Vong, S. & Kalluri, R. The role of stromal myofibroblast and extracellular matrix in tumor angiogenesis. Genes Cancer 2, 1139–1145 (2011).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013). A good review of the tumour microenvironment.
Kalluri, R. Basement membranes: structure, assembly and role in tumour angiogenesis. Nat. Rev. Cancer 3, 422–433 (2003).
Hanahan, D. & Coussens, L. M. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21, 309–322 (2012).
Pietras, K. & Ostman, A. Hallmarks of cancer: interactions with the tumor stroma. Exp. Cell Res. 316, 1324–1331 (2010).
Bhowmick, N. A. et al. TGF-β signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science 303, 848–851 (2004).
Lujambio, A. et al. Non-cell-autonomous tumor suppression by p53. Cell 153, 449–460 (2013).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Kalluri, R. & Zeisberg, M. Fibroblasts in cancer. Nat. Rev. Cancer 6, 392–401 (2006).
Ohlund, D., Elyada, E. & Tuveson, D. Fibroblast heterogeneity in the cancer wound. J. Exp. Med. 211, 1503–1523 (2014). A review about CAFs and their likely heterogeneity.
Marsh, T., Pietras, K. & McAllister, S. S. Fibroblasts as architects of cancer pathogenesis. Biochim. Biophys. Acta 1832, 1070–1078 (2013).
Ostman, A. & Augsten, M. Cancer-associated fibroblasts and tumor growth—bystanders turning into key players. Curr. Opin. Genet. Dev. 19, 67–73 (2009).
Tampe, B. & Zeisberg, M. Contribution of genetics and epigenetics to progression of kidney fibrosis. Nephrol. Dial. Transpl. 29 (Suppl. 4), iv72–iv79 (2013).
Zeisberg, E. M. & Zeisberg, M. The role of promoter hypermethylation in fibroblast activation and fibrogenesis. J. Pathol. 229, 264–273 (2013).
Micallef, L. et al. The myofibroblast, multiple origins for major roles in normal and pathological tissue repair. Fibrogenesis Tissue Repair 5, S5 (2012).
Ronnov-Jessen, L. & Petersen, O. W. Induction of α-smooth muscle actin by transforming growth factor-β1 in quiescent human breast gland fibroblasts. Implications for myofibroblast generation in breast neoplasia. Lab. Invest. 68, 696–707 (1993).
Paunescu, V. et al. Tumour-associated fibroblasts and mesenchymal stem cells: more similarities than differences. J. Cell. Mol. Med. 15, 635–646 (2011). A comprehensive comparative analysis of MSCs and CAFs.
Sappino, A. P., Skalli, O., Jackson, B., Schurch, W. & Gabbiani, G. Smooth-muscle differentiation in stromal cells of malignant and non-malignant breast tissues. Int. J. Cancer 41, 707–712 (1988).
Powell, D. W. et al. Myofibroblasts. I. Paracrine cells important in health and disease. Am. J. Physiol. 277, C1–C9 (1999).
LeBleu, V. S. et al. Origin and function of myofibroblasts in kidney fibrosis. Nat. Med. 19, 1047–1053 (2013).
Zeisberg, M., Strutz, F. & Muller, G. A. Role of fibroblast activation in inducing interstitial fibrosis. J. Nephrol. 13 (Suppl. 3), S111–S120 (2000).
Desmouliere, A., Darby, I. A. & Gabbiani, G. Normal and pathologic soft tissue remodeling: role of the myofibroblast, with special emphasis on liver and kidney fibrosis. Lab. Invest. 83, 1689–1707 (2003).
Lajiness, J. D. & Conway, S. J. The dynamic role of cardiac fibroblasts in development and disease. J. Cardiovasc. Transl Res. 5, 739–748 (2012).
Dvorak, H. F. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N. Engl. J. Med. 315, 1650–1659 (1986).
Monaco, J. L. & Lawrence, W. T. Acute wound healing an overview. Clin. Plast. Surg. 30, 1–12 (2003).
Gurtner, G. C., Werner, S., Barrandon, Y. & Longaker, M. T. Wound repair and regeneration. Nature 453, 314–321 (2008).
Bliss, L. A. et al. Use of postmortem human dura mater and scalp for deriving human fibroblast cultures. PLoS One 7, e45282 (2012).
Virchow, R. Die Cellularpathologie in lhrer Begruendung auf Physiologische und Pathologische Gewebelehre (ed. Hirschwald, A.) (Berlin, 1858).
Duvall, M. Atlas d'Embryologie. (ed. Masson, G.) (Paris, 1879).
Tarin, D. & Croft, C. B. Ultrastructural features of wound healing in mouse skin. J. Anat. 105, 189–190 (1969).
Gabbiani, G., Ryan, G. B. & Majne, G. Presence of modified fibroblasts in granulation tissue and their possible role in wound contraction. Experientia 27, 549–550 (1971).
Darby, I. A., Laverdet, B., Bonte, F. & Desmouliere, A. Fibroblasts and myofibroblasts in wound healing. Clin. Cosmet. Investigat. Dermatol. 7, 301–311 (2014).
Castor, C. W., Wilson, S. M., Heiss, P. R. & Seidman, J. C. Activation of lung connective tissue cells in vitro. Am. Rev. Respir. Dis. 120, 101–106 (1979).
Muller, G. A. & Rodemann, H. P. Characterization of human renal fibroblasts in health and disease: I. Immunophenotyping of cultured tubular epithelial cells and fibroblasts derived from kidneys with histologically proven interstitial fibrosis. Am. J. Kidney Dis. 17, 680–683 (1991).
Tomasek, J. J., Gabbiani, G., Hinz, B., Chaponnier, C. & Brown, R. A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 3, 349–363 (2002).
Parsonage, G. et al. A stromal address code defined by fibroblasts. Trends Immunol. 26, 150–156 (2005).
Karnoub, A. E. et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449, 557–563 (2007). A study showing that MSC-derived CCL5 enhances breast cancer metastasis.
Quante, M. et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 19, 257–272 (2011). A study indicating that MSC-derived CAFs promote inflammation-induced gastric cancer progression and are characterized by global DNA hypomethylation and elevated expression of IL-6, WNT5A and bone morphogenetic protein 4 (BMP4).
Xu, J. et al. Contribution of bone marrow-derived fibrocytes to liver fibrosis. Hepatobiliary Surg. Nutr. 4, 34–47 (2015).
Raab, S., Klingenstein, M., Liebau, S. & Linta, L. A. Comparative view on human somatic cell sources for iPSC generation. Stem Cells Int. 2014, 768391 (2014).
Lorenz, K. et al. Multilineage differentiation potential of human dermal skin-derived fibroblasts. Exp. Dermatol. 17, 925–932 (2008).
Miyake, T. & Kalluri, R. Cardiac biology: cell plasticity helps hearts to repair. Nature 514, 575–576 (2014).
Ubil, E. et al. Mesenchymal-endothelial transition contributes to cardiac neovascularization. Nature 514, 585–590 (2014).
Sriram, G., Bigliardi, P. L. & Bigliardi-Qi, M. Fibroblast heterogeneity and its implications for engineering organotypic skin models in vitro. Eur. J. Cell Biol. 94, 483–512 (2015).
Driskell, R. R. & Watt, F. M. Understanding fibroblast heterogeneity in the skin. Trends Cell Biol. 25, 92–99 (2015).
Rodemann, H. P. & Muller, G. A. Characterization of human renal fibroblasts in health and disease: II. In vitro growth, differentiation, and collagen synthesis of fibroblasts from kidneys with interstitial fibrosis. Am. J. Kidney Dis. 17, 684–686 (1991).
Simian, M. et al. The interplay of matrix metalloproteinases, morphogens and growth factors is necessary for branching of mammary epithelial cells. Development 128, 3117–3131 (2001).
Wiseman, B. S. & Werb, Z. Stromal effects on mammary gland development and breast cancer. Science 296, 1046–1049 (2002).
Driskell, R. R. et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504, 277–281 (2013).
Dulauroy, S., Di Carlo, S. E., Langa, F., Eberl, G. & Peduto, L. Lineage tracing and genetic ablation of ADAM12+ perivascular cells identify a major source of profibrotic cells during acute tissue injury. Nat. Med. 18, 1262–1270 (2012).
Hamburg-Shields, E., DiNuoscio, G. J., Mullin, N. K., Lafyatis, R. & Atit, R. P. Sustained β-catenin activity in dermal fibroblasts promotes fibrosis by up-regulating expression of extracellular matrix protein-coding genes. J. Pathol. 235, 686–697 (2015).
Rock, J. R. et al. Multiple stromal populations contribute to pulmonary fibrosis without evidence for epithelial to mesenchymal transition. Proc. Natl Acad. Sci. USA 108, E1475–E1483 (2011).
De Wever, O., Van Bockstal, M., Mareel, M., Hendrix, A. & Bracke, M. Carcinoma-associated fibroblasts provide operational flexibility in metastasis. Semin. Cancer Biol. 25, 33–46 (2014).
Dumont, N. et al. Breast fibroblasts modulate early dissemination, tumorigenesis, and metastasis through alteration of extracellular matrix characteristics. Neoplasia 15, 249–262 (2013).
Ryan, G. B. et al. Myofibroblasts in an avascular fibrous tissue. Lab. Invest. 29, 197–206 (1973).
Ryan, G. B. et al. Myofibroblasts in human granulation tissue. Hum. Pathol. 5, 55–67 (1974).
Tsukada, T., McNutt, M. A., Ross, R. & Gown, A. M. HHF35, a muscle actin-specific monoclonal antibody. II. Reactivity in normal, reactive, and neoplastic human tissues. Am. J. Pathol. 127, 389–402 (1987).
Schor, S. L., Schor, A. M., Grey, A. M. & Rushton, G. Foetal and cancer patient fibroblasts produce an autocrine migration-stimulating factor not made by normal adult cells. J. Cell Sci. 90, 391–399 (1988).
Durning, P., Schor, S. L. & Sellwood, R. A. Fibroblasts from patients with breast cancer show abnormal migratory behaviour in vitro. Lancet 2, 890–892 (1984).
Elenbaas, B. & Weinberg, R. A. Heterotypic signaling between epithelial tumor cells and fibroblasts in carcinoma formation. Exp. Cell Res. 264, 169–184 (2001).
Lohr, M. et al. Transforming growth factor-β1 induces desmoplasia in an experimental model of human pancreatic carcinoma. Cancer Res. 61, 550–555 (2001).
Aoyagi, Y. et al. Overexpression of TGF-β by infiltrated granulocytes correlates with the expression of collagen mRNA in pancreatic cancer. Br. J. Cancer 91, 1316–1326 (2004).
Ishii, G., Ochiai, A. & Neri, S. Phenotypic and functional heterogeneity of cancer-associated fibroblast within the tumor microenvironment. Adv. Drug Delivery Rev. 99, 186–196 (2015).
Bronzert, D. A. et al. Synthesis and secretion of platelet-derived growth factor by human breast cancer cell lines. Proc. Natl Acad. Sci. USA 84, 5763–5767 (1987).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Dvorak, H. F., Form, D. M., Manseau, E. J. & Smith, B. D. Pathogenesis of desmoplasia. I. Immunofluorescence identification and localization of some structural proteins of line 1 and line 10 guinea pig tumors and of healing wounds. J. Natl Cancer Inst. 73, 1195–1205 (1984).
Folkman, J. & Kalluri, R. Cancer without disease. Nature 427, 787 (2004). A concept piece proposing the cancer-restraining actions of desmoplasia and that cancer can exist without resulting in clinical disease.
Polyak, K. & Kalluri, R. The role of the microenvironment in mammary gland development and cancer. Cold Spring Harb. Perspect. Biol. 2, a003244 (2010).
Erez, N., Truitt, M., Olson, P., Arron, S. T. & Hanahan, D. Cancer-associated fibroblasts are activated in incipient neoplasia to orchestrate tumor-promoting inflammation in an NF-κB-dependent manner. Cancer Cell 17, 135–147 (2010). An interesting study indicating that CAFs aquire a pro-inflammatory gene expression programme in the early stages of neoplasia.
Dolberg, D. S., Hollingsworth, R., Hertle, M. & Bissell, M. J. Wounding and its role in RSV-mediated tumor formation. Science 230, 676–678 (1985).
Schuh, A. C., Keating, S. J., Monteclaro, F. S., Vogt, P. K. & Breitman, M. L. Obligatory wounding requirement for tumorigenesis in v-jun transgenic mice. Nature 346, 756–760 (1990).
Li, J. et al. Idiopathic pulmonary fibrosis will increase the risk of lung cancer. Chinese Med. J. 127, 3142–3149 (2014).
Park, J. et al. Lung cancer in patients with idiopathic pulmonary fibrosis. Eur. Respir. J. 17, 1216–1219 (2001).
Samet, J. M. Does idiopathic pulmonary fibrosis increase lung cancer risk? Am. J. Respir. Crit. Care Med. 161, 1–2 (2000).
Sangiovanni, A. et al. Increased survival of cirrhotic patients with a hepatocellular carcinoma detected during surveillance. Gastroenterology 126, 1005–1014 (2004).
Wang, H. M. et al. Liver stiffness measurement as an alternative to fibrotic stage in risk assessment of hepatocellular carcinoma incidence for chronic hepatitis C patients. Liver Int. 33, 756–761 (2013).
Fukumura, D. et al. Tumor induction of VEGF promoter activity in stromal cells. Cell 94, 715–725 (1998).
Brown, L. F. et al. Vascular stroma formation in carcinoma in situ, invasive carcinoma, and metastatic carcinoma of the breast. Clin. Cancer Res. 5, 1041–1056 (1999).
Feng, D. et al. Ultrastructural localization of the vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) receptor-2 (FLK-1, KDR) in normal mouse kidney and in the hyperpermeable vessels induced by VPF/VEGF-expressing tumors and adenoviral vectors. J. Histochem. Cytochem. 48, 545–556 (2000).
Leung, D. W., Cachianes, G., Kuang, W. J., Goeddel, D. V. & Ferrara, N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246, 1306–1309 (1989).
Giussani, M., Merlino, G., Cappelletti, V., Tagliabue, E. & Daidone, M. G. Tumor–extracellular matrix interactions: Identification of tools associated with breast cancer progression. Semin. Cancer Biol. 35, 3–10 (2015).
Chiquet-Ehrismann, R., Mackie, E. J., Pearson, C. A. & Sakakura, T. Tenascin: an extracellular matrix protein involved in tissue interactions during fetal development and oncogenesis. Cell 47, 131–139 (1986).
Mackie, E. J. et al. Tenascin is a stromal marker for epithelial malignancy in the mammary gland. Proc. Natl Acad. Sci. USA 84, 4621–4625 (1987).
Kyutoku, M. et al. Role of periostin in cancer progression and metastasis: inhibition of breast cancer progression and metastasis by anti-periostin antibody in a murine model. Int. J. Mol. Med. 28, 181–186 (2011).
Ruan, K., Bao, S. & Ouyang, G. The multifaceted role of periostin in tumorigenesis. Cell. Mol. Life Sci. 66, 2219–2230 (2009).
Abdollahi, A. et al. Inhibition of platelet-derived growth factor signaling attenuates pulmonary fibrosis. J. Exp. Med. 201, 925–935 (2005).
Pietras, K., Pahler, J., Bergers, G. & Hanahan, D. Functions of paracrine PDGF signaling in the proangiogenic tumor stroma revealed by pharmacological targeting. PLoS Med. 5, e19 (2008).
Paulsson, J., Ehnman, M. & Ostman, A. PDGF receptors in tumor biology: prognostic and predictive potential. Future Oncol. 10, 1695–1708 (2014).
Strutz, F. et al. Identification and characterization of a fibroblast marker: FSP1. J. Cell Biol. 130, 393–405 (1995).
Osterreicher, C. H. et al. Fibroblast-specific protein 1 identifies an inflammatory subpopulation of macrophages in the liver. Proc. Natl Acad. Sci. USA 108, 308–313 (2011).
Kikuchi, N. et al. Nuclear expression of S100A4 is associated with aggressive behavior of epithelial ovarian carcinoma: an important autocrine/paracrine factor in tumor progression. Cancer Sci. 97, 1061–1069 (2006).
Arnold, J. N., Magiera, L., Kraman, M. & Fearon, D. T. Tumoral immune suppression by macrophages expressing fibroblast activation protein-α and heme oxygenase-1. Cancer Immunol. Res. 2, 121–126 (2014).
Armulik, A., Genove, G. & Betsholtz, C. Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev. Cell 21, 193–215 (2011).
Ozdemir, B. C. et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell 25, 719–734 (2014). This paper shows that depletion of α SMA+ stromal cells promotes an immunosupressive tumour milieu and exacerbates cancer progression with diminished survival.
Sugimoto, H., Mundel, T. M., Kieran, M. W. & Kalluri, R. Identification of fibroblast heterogeneity in the tumor microenvironment. Cancer Biol. Ther. 5, 1640–1646 (2006).
Guido, C. et al. Metabolic reprogramming of cancer-associated fibroblasts by TGF-β drives tumor growth: connecting TGF-β signaling with “Warburg-like” cancer metabolism and L-lactate production. Cell Cycle 11, 3019–3035 (2012).
Simpkins, S. A., Hanby, A. M., Holliday, D. L. & Speirs, V. Clinical and functional significance of loss of caveolin-1 expression in breast cancer-associated fibroblasts. J. Pathol. 227, 490–498 (2012).
Goetz, J. G. et al. Biomechanical remodeling of the microenvironment by stromal caveolin-1 favors tumor invasion and metastasis. Cell 146, 148–163 (2011).
Li, P. et al. Epigenetic silencing of microRNA-149 in cancer-associated fibroblasts mediates prostaglandin E2/interleukin-6 signaling in the tumor microenvironment. Cell Res. 25, 588–603 (2015).
Mrazek, A. A. et al. Colorectal cancer-associated fibroblasts are genotypically distinct. Curr. Cancer Ther. Rev. 10, 97–218 (2014).
Hu, M. et al. Distinct epigenetic changes in the stromal cells of breast cancers. Nat. Genet. 37, 899–905 (2005).
Bechtel, W. et al. Methylation determines fibroblast activation and fibrogenesis in the kidney. Nat. Med. 16, 544–550 (2010). The first study to implicate epigenetic control of fibroblast activation.
Huang, S. K. et al. Histone modifications are responsible for decreased Fas expression and apoptosis resistance in fibrotic lung fibroblasts. Cell Death Dis. 4, e621 (2013).
He, Z. et al. Epigenetic regulation of Thy-1 gene expression by histone modification is involved in lipopolysaccharide-induced lung fibroblast proliferation. J. Cell. Mol. Med. 17, 160–167 (2013).
Robinson, C. M., Neary, R., Levendale, A., Watson, C. J. & Baugh, J. A. Hypoxia-induced DNA hypermethylation in human pulmonary fibroblasts is associated with Thy-1 promoter methylation and the development of a pro-fibrotic phenotype. Respir. Res. 13, 74 (2012).
Zong, Y. et al. Stromal epigenetic dysregulation is sufficient to initiate mouse prostate cancer via paracrine Wnt signaling. Proc. Natl Acad. Sci. USA 109, E3395–E3404 (2012). This study shows that overexpression of the chromatin remodeller HMGA2 in stroma initiates neoplasia of prostate epithelium.
Albrengues, J. et al. Epigenetic switch drives the conversion of fibroblasts into proinvasive cancer-associated fibroblasts. Nat. Commun. 6, 10204 (2015).
Madar, S. et al. Modulated expression of WFDC1 during carcinogenesis and cellular senescence. Carcinogenesis 30, 20–27 (2009).
Orimo, A. et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121, 335–348 (2005).
Olumi, A. F. et al. Carcinoma-associated fibroblasts direct tumor progression of initiated human prostatic epithelium. Cancer Res. 59, 5002–5011 (1999).
Dimanche-Boitrel, M. T. et al. In vivo and in vitro invasiveness of a rat colon-cancer cell line maintaining E-cadherin expression: an enhancing role of tumor-associated myofibroblasts. Int. J. Cancer 56, 512–521 (1994).
Scherz-Shouval, R. et al. The reprogramming of tumor stroma by HSF1 is a potent enabler of malignancy. Cell 158, 564–578 (2014).
Calvo, F. et al. Mechanotransduction and YAP-dependent matrix remodelling is required for the generation and maintenance of cancer-associated fibroblasts. Nat. Cell Biol. 15, 637–646 (2013).
Procopio, M. G. et al. Combined CSL and p53 downregulation promotes cancer-associated fibroblast activation. Nat. Cell Biol. 17, 1193–1204 (2015).
Boire, A. et al. PAR1 is a matrix metalloprotease-1 receptor that promotes invasion and tumorigenesis of breast cancer cells. Cell 120, 303–313 (2005).
Stetler-Stevenson, W. G., Aznavoorian, S. & Liotta, L. A. Tumor cell interactions with the extracellular matrix during invasion and metastasis. Annu. Rev. Cell Biol. 9, 541–573 (1993).
Sternlicht, M. D. et al. The stromal proteinase MMP3/stromelysin-1 promotes mammary carcinogenesis. Cell 98, 137–146 (1999).
Lochter, A. et al. Matrix metalloproteinase stromelysin-1 triggers a cascade of molecular alterations that leads to stable epithelial-to-mesenchymal conversion and a premalignant phenotype in mammary epithelial cells. J. Cell Biol. 139, 1861–1872 (1997).
Borges, F. T. et al. TGF-β1-containing exosomes from injured epithelial cells activate fibroblasts to initiate tissue regenerative responses and fibrosis. J. Am. Soc. Nephrol. 24, 385–392 (2013).
Kahlert, C. & Kalluri, R. Exosomes in tumor microenvironment influence cancer progression and metastasis. J. Mol. Med. (Berl.) 91, 431–437 (2013).
Luga, V. & Wrana, J. L. Tumor–stroma interaction: revealing fibroblast-secreted exosomes as potent regulators of Wnt-planar cell polarity signaling in cancer metastasis. Cancer Res. 73, 6843–6847 (2013).
Hu, Y. et al. Fibroblast-derived exosomes contribute to chemoresistance through priming cancer stem cells in colorectal cancer. PLoS One 10, e0125625 (2015).
Shimoda, M. et al. Loss of the Timp gene family is sufficient for the acquisition of the CAF-like cell state. Nat. Cell Biol. 16, 889–901 (2014).
Malanchi, I. et al. Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481, 85–89 (2012).
Vermeulen, L. et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat. Cell Biol. 12, 468–476 (2010).
Del Pozo Martin, Y. et al. Mesenchymal cancer cell-stroma crosstalk promotes niche activation, epithelial reversion, and metastatic colonization. Cell Rep. 13, 2456–2469 (2015).
Chen, W. J. et al. Cancer-associated fibroblasts regulate the plasticity of lung cancer stemness via paracrine signalling. Nat. Commun. 5, 3472 (2014).
Elkabets, M. et al. Human tumors instigate granulin-expressing hematopoietic cells that promote malignancy by activating stromal fibroblasts in mice. J. Clin. Invest. 121, 784–799 (2011).
Bruzzese, F. et al. Local and systemic protumorigenic effects of cancer-associated fibroblast-derived GDF15. Cancer Res. 74, 3408–3417 (2014).
Levental, K. R. et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139, 891–906 (2009).
Gaggioli, C. et al. Fibroblast-led collective invasion of carcinoma cells with differing roles for RhoGTPases in leading and following cells. Nat. Cell Biol. 9, 1392–1400 (2007). This paper reports ECM remodelling by CAFs to engineer tracks used by cancer cells to migrate.
O'Connell, J. T. et al. VEGF-A and Tenascin-C produced by S100A4+ stromal cells are important for metastatic colonization. Proc. Natl Acad. Sci. USA 108, 16002–16007 (2011). This study reports that FSP1+ stromal cells remodel the metastatic soil.
Olaso, E. et al. Tumor-dependent activation of rodent hepatic stellate cells during experimental melanoma metastasis. Hepatology 26, 634–642 (1997).
Calon, A. et al. Dependency of colorectal cancer on a TGF-β-driven program in stromal cells for metastasis initiation. Cancer Cell 22, 571–584 (2012).
Pena, C. et al. STC1 expression by cancer-associated fibroblasts drives metastasis of colorectal cancer. Cancer Res. 73, 1287–1297 (2013).
Kaplan, R. N. et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 438, 820–827 (2005).
Grum-Schwensen, B. et al. Suppression of tumor development and metastasis formation in mice lacking the S100A4(mts1) gene. Cancer Res. 65, 3772–3780 (2005).
Vander Heiden, M. G., Cantley, L. C. & Thompson, C. B. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 324, 1029–1033 (2009).
Martinez-Outschoorn, U. E., Lisanti, M. P. & Sotgia, F. Catabolic cancer-associated fibroblasts transfer energy and biomass to anabolic cancer cells, fueling tumor growth. Semin. Cancer Biol. 25, 47–60 (2014).
Zhang, D. et al. Metabolic reprogramming of cancer-associated fibroblasts by IDH3α downregulation. Cell Rep. 10, 1335–1348 (2015).
Chaudhri, V. K. et al. Metabolic alterations in lung cancer-associated fibroblasts correlated with increased glycolytic metabolism of the tumor. Mol. Cancer Res. 11, 579–592 (2013).
Pavlides, S. et al. The reverse Warburg effect: aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle 8, 3984–4001 (2009). This study indicates that Cav1 -knockout fibroblasts metabolically cooperate with cancer cells.
LeBleu, V. S. et al. PGC-1α mediates mitochondrial biogenesis and oxidative phosphorylation in cancer cells to promote metastasis. Nat. Cell Biol. 16, 992–1003, 1001–1015 (2014).
Ying, H. et al. Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell 149, 656–670 (2012).
Viale, A. et al. Oncogene ablation-resistant pancreatic cancer cells depend on mitochondrial function. Nature 514, 628–632 (2014).
Fiaschi, T. et al. Reciprocal metabolic reprogramming through lactate shuttle coordinately influences tumor-stroma interplay. Cancer Res. 72, 5130–5140 (2012).
Valencia, T. et al. Metabolic reprogramming of stromal fibroblasts through p62-mTORC1 signaling promotes inflammation and tumorigenesis. Cancer Cell 26, 121–135 (2014).
Kuchnio, A. et al. The cancer cell oxygen sensor PHD2 promotes metastasis via activation of cancer-associated fibroblasts. Cell Rep. 12, 992–1005 (2015).
Madsen, C. D. et al. Hypoxia and loss of PHD2 inactivate stromal fibroblasts to decrease tumour stiffness and metastasis. EMBO Rep. 16, 1394–1408 (2015).
Chang, C. H. et al. Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell 162, 1229–1241 (2015). This paper implicates PDL1 in the control of metabolic competition in the TME to mediate T cell hyper-responsiveness.
Ghesquiere, B., Wong, B. W., Kuchnio, A. & Carmeliet, P. Metabolism of stromal and immune cells in health and disease. Nature 511, 167–176 (2014).
Molon, B., Cali, B. & Viola, A. T. Cells and cancer: how metabolism shapes immunity. Front. Immunol. 7, 20 (2016).
Turley, S. J., Cremasco, V. & Astarita, J. L. Immunological hallmarks of stromal cells in the tumour microenvironment. Nat. Rev. Immunol. 15, 669–682 (2015).
Lisanti, M. P., Martinez-Outschoorn, U. E. & Sotgia, F. Oncogenes induce the cancer-associated fibroblast phenotype: metabolic symbiosis and “fibroblast addiction” are new therapeutic targets for drug discovery. Cell Cycle 12, 2723–2732 (2013).
Costea, D. E. et al. Identification of two distinct carcinoma-associated fibroblast subtypes with differential tumor-promoting abilities in oral squamous cell carcinoma. Cancer Res. 73, 3888–3901 (2013).
De Boeck, A. et al. Differential secretome analysis of cancer-associated fibroblasts and bone marrow-derived precursors to identify microenvironmental regulators of colon cancer progression. Proteomics 13, 379–388 (2013).
Koczorowska, M. M. et al. Fibroblast activation protein-α, a stromal cell surface protease, shapes key features of cancer associated fibroblasts through proteome and degradome alterations. Mol. Oncol. 10, 40–58 (2015).
Lotti, F. et al. Chemotherapy activates cancer-associated fibroblasts to maintain colorectal cancer-initiating cells by IL-17A. J. Exp. Med. 210, 2851–2872 (2013).
Raffaghello, L. & Dazzi, F. Classification and biology of tumour associated stromal cells. Immunol. Lett. 168, 175–182 (2015).
Harper, J. & Sainson, R. C. Regulation of the anti-tumour immune response by cancer-associated fibroblasts. Semin. Cancer Biol. 25, 69–77 (2014).
Patel, R., Filer, A., Barone, F. & Buckley, C. D. Stroma: fertile soil for inflammation. Best practice and research. Clin. Rheumatol. 28, 565–576 (2014).
Poggi, A., Musso, A., Dapino, I. & Zocchi, M. R. Mechanisms of tumor escape from immune system: role of mesenchymal stromal cells. Immunol. Lett. 159, 55–72 (2014).
Soleymaninejadian, E., Pramanik, K. & Samadian, E. Immunomodulatory properties of mesenchymal stem cells: cytokines and factors. Am. J. Reprod. Immunol. 67, 1–8 (2012).
Park, S. J. et al. IL-6 regulates in vivo dendritic cell differentiation through STAT3 activation. J. Immunol. 173, 3844–3854 (2004).
Chomarat, P., Banchereau, J., Davoust, J. & Palucka, A. K. IL-6 switches the differentiation of monocytes from dendritic cells to macrophages. Nat. Immunol. 1, 510–514 (2000).
Hugo, H. J. et al. Contribution of fibroblast and mast cell (afferent) and tumor (efferent) IL-6 effects within the tumor microenvironment. Cancer Microenviron. 5, 83–93 (2012).
Wan, Y. Y. & Flavell, R. A. 'Yin-Yang' functions of transforming growth factor-β and T regulatory cells in immune regulation. Immunol. Rev. 220, 199–213 (2007).
Bailey, S. R. et al. Th17 cells in cancer: the ultimate identity crisis. Front. Immunol. 5, 276 (2014). A detailed review on T H 17 cells in the TME.
Li, M. O., Wan, Y. Y., Sanjabi, S., Robertson, A. K. & Flavell, R. A. Transforming growth factor-β regulation of immune responses. Annu. Rev. Immunol. 24, 99–146 (2006).
Kim, J. H. et al. The role of myofibroblasts in upregulation of S100A8 and S100A9 and the differentiation of myeloid cells in the colorectal cancer microenvironment. Biochem. Biophys. Res. Commun. 423, 60–66 (2012).
Augsten, M. et al. Cancer-associated fibroblasts expressing CXCL14 rely upon NOS1-derived nitric oxide signaling for their tumor-supporting properties. Cancer Res. 74, 2999–3010 (2014).
Mishra, P., Banerjee, D. & Ben-Baruch, A. Chemokines at the crossroads of tumor-fibroblast interactions that promote malignancy. J. Leukoc. Biol. 89, 31–39 (2011).
Van Linthout, S., Miteva, K. & Tschope, C. Crosstalk between fibroblasts and inflammatory cells. Cardiovasc. Res. 102, 258–269 (2014).
Qian, B. Z. et al. CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475, 222–225 (2011).
Silzle, T. et al. Tumor-associated fibroblasts recruit blood monocytes into tumor tissue. Eur. J. Immunol. 33, 1311–1320 (2003).
Barnas, J. L., Simpson-Abelson, M. R., Yokota, S. J., Kelleher, R. J. & Bankert, R. B. T cells and stromal fibroblasts in human tumor microenvironments represent potential therapeutic targets. Cancer Microenviron. 3, 29–47 (2010).
Pinchuk, I. V. et al. PD-1 ligand expression by human colonic myofibroblasts/fibroblasts regulates CD4+ T-cell activity. Gastroenterology 135, 1228–1237 (2008).
Nazareth, M. R. et al. Characterization of human lung tumor-associated fibroblasts and their ability to modulate the activation of tumor-associated T cells. J. Immunol. 178, 5552–5562 (2007).
Augsten, M. Cancer-associated fibroblasts as another polarized cell type of the tumor microenvironment. Front. Oncol. 4, 62 (2014).
Salmon, H. et al. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J. Clin. Invest. 122, 899–910 (2012).
Egeblad, M. & Werb, Z. New functions for the matrix metalloproteinases in cancer progression. Nat. Rev. Cancer 2, 161–174 (2002).
Kraman, M. et al. Suppression of antitumor immunity by stromal cells expressing fibroblast activation protein-α. Science 330, 827–830 (2010). This article shows that depletion of FAP+ stromal cells enables immunological control of tumour growth.
Wen, Y. et al. Immunotherapy targeting fibroblast activation protein inhibits tumor growth and increases survival in a murine colon cancer model. Cancer Sci. 101, 2325–2332 (2010).
Liao, D., Luo, Y., Markowitz, D., Xiang, R. & Reisfeld, R. A. Cancer associated fibroblasts promote tumor growth and metastasis by modulating the tumor immune microenvironment in a 4T1 murine breast cancer model. PLoS One 4, e7965 (2009).
Ohshio, Y. et al. Cancer-associated fibroblast-targeted strategy enhances antitumor immune responses in dendritic cell-based vaccine. Cancer Sci. 106, 134–142 (2015).
Rhim, A. D. et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell 25, 735–747 (2014).
Fidler, I. J. et al. Modulation of tumor cell response to chemotherapy by the organ environment. Cancer Metastasis Rev. 13, 209–222 (1994).
Farmer, P. et al. A stroma-related gene signature predicts resistance to neoadjuvant chemotherapy in breast cancer. Nat. Med. 15, 68–74 (2009).
Meads, M. B., Gatenby, R. A. & Dalton, W. S. Environment-mediated drug resistance: a major contributor to minimal residual disease. Nat. Rev. Cancer 9, 665–674 (2009). A review on the role of the TME in drug resistance.
Paraiso, K. H. & Smalley, K. S. Fibroblast-mediated drug resistance in cancer. Biochem. Pharmacol. 85, 1033–1041 (2013).
Heldin, C. H., Rubin, K., Pietras, K. & Ostman, A. High interstitial fluid pressure - an obstacle in cancer therapy. Nat. Rev. Cancer 4, 806–813 (2004).
Correia, A. L. & Bissell, M. J. The tumor microenvironment is a dominant force in multidrug resistance. Drug Resist. Updat. 15, 39–49 (2012).
Dittmer, J. & Leyh, B. The impact of tumor stroma on drug response in breast cancer. Semin. Cancer Biol. 31, 3–15 (2015).
Mori, Y. et al. Anti-alpha4 integrin antibody suppresses the development of multiple myeloma and associated osteoclastic osteolysis. Blood 104, 2149–2154 (2004).
Park, C. C., Zhang, H. J., Yao, E. S., Park, C. J. & Bissell, M. J. β1 integrin inhibition dramatically enhances radiotherapy efficacy in human breast cancer xenografts. Cancer Res. 68, 4398–4405 (2008).
Hazlehurst, L. A., Damiano, J. S., Buyuksal, I., Pledger, W. J. & Dalton, W. S. Adhesion to fibronectin via β1 integrins regulates p27kip1 levels and contributes to cell adhesion mediated drug resistance (CAM-DR). Oncogene 19, 4319–4327 (2000).
White, D. E., Rayment, J. H. & Muller, W. J. Addressing the role of cell adhesion in tumor cell dormancy. Cell Cycle 5, 1756–1759 (2006).
Hirata, E. et al. Intravital imaging reveals how BRAF inhibition generates drug-tolerant microenvironments with high integrin β1/FAK signaling. Cancer Cell 27, 574–588 (2015).
Flach, E. H., Rebecca, V. W., Herlyn, M., Smalley, K. S. & Anderson, A. R. Fibroblasts contribute to melanoma tumor growth and drug resistance. Mol. Pharmaceut. 8, 2039–2049 (2011).
Li, G., Satyamoorthy, K. & Herlyn, M. N-Cadherin-mediated intercellular interactions promote survival and migration of melanoma cells. Cancer Res. 61, 3819–3825 (2001).
Sui, H., Zhu, L., Deng, W. & Li, Q. Epithelial-mesenchymal transition and drug resistance: role, molecular mechanisms, and therapeutic strategies. Oncol. Res. Treat. 37, 584–589 (2014).
Mitra, A., Mishra, L. & Li, S. EMT, CTCs and CSCs in tumor relapse and drug-resistance. Oncotarget 6, 10697–10711 (2015).
Chang, J. T. & Mani, S. A. Sheep, wolf, or werewolf: cancer stem cells and the epithelial-to-mesenchymal transition. Cancer Lett. 341, 16–23 (2013).
Zheng, X. et al. Epithelial-to-mesenchymal transition is dispensable for metastasis but induces chemoresistance in pancreatic cancer. Nature 527, 525–530 (2015).
Kumari, N., Dwarakanath, B. S., Das, A. & Bhatt, A. N. Role of interleukin-6 in cancer progression and therapeutic resistance. Tumour Biol. http://dx.doi.org/10.1007/s13277-016-5098-7 (2016).
Wilson, T. R. et al. Widespread potential for growth-factor-driven resistance to anticancer kinase inhibitors. Nature 487, 505–509 (2012).
Straussman, R. et al. Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 487, 500–504 (2012).
Wang, W. et al. Crosstalk to stromal fibroblasts induces resistance of lung cancer to epidermal growth factor receptor tyrosine kinase inhibitors. Clin. Cancer Res. 15, 6630–6638 (2009).
McMillin, D. W., Negri, J. M. & Mitsiades, C. S. The role of tumour-stromal interactions in modifying drug response: challenges and opportunities. Nat. Rev. Drug Discov. 12, 217–228 (2013).
Olive, K. P. et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 324, 1457–1461 (2009).
Madden, J. I. Infinity reports update from phase 2 study of saridegib plus gemcitabine in patients with metastatic pancreatic cancer. Infinity Pharmaceuticals http://phx.corporate-ir.net/phoenix.zhtml?c=121941&p=irol-newsArticle&ID=1653550 (27 January 2012).
Jacobetz, M. A. et al. Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer. Gut 62, 112–120 (2013).
Provenzano, P. P. et al. Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 21, 418–429 (2012).
Goel, S., Wong, A. H. & Jain, R. K. Vascular normalization as a therapeutic strategy for malignant and nonmalignant disease. Cold Spring Harb. Perspect. Med. 2, a006486 (2012).
Huang, Y., Goel, S., Duda, D. G., Fukumura, D. & Jain, R. K. Vascular normalization as an emerging strategy to enhance cancer immunotherapy. Cancer Res. 73, 2943–2948 (2013).
Kuperwasser, C. et al. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc. Natl Acad. Sci. USA 101, 4966–4971 (2004).
Cheng, N. et al. Loss of TGF-β type II receptor in fibroblasts promotes mammary carcinoma growth and invasion through upregulation of TGF-α-, MSP- and HGF-mediated signaling networks. Oncogene 24, 5053–5068 (2005).
Lewis, D. A., Travers, J. B., Machado, C., Somani, A. K. & Spandau, D. F. Reversing the aging stromal phenotype prevents carcinoma initiation. Aging 3, 407–416 (2011).
Paulsson, J. & Micke, P. Prognostic relevance of cancer-associated fibroblasts in human cancer. Semin. Cancer Biol. 25, 61–68 (2014).
Wang, W. Q. et al. Intratumoral α-SMA enhances the prognostic potency of CD34 associated with maintenance of microvessel integrity in hepatocellular carcinoma and pancreatic cancer. PLoS One 8, e71189 (2013).
Finak, G. et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat. Med. 14, 518–527 (2008).
Frings, O. et al. Prognostic significance in breast cancer of a gene signature capturing stromal PDGF signaling. Am. J. Pathol. 182, 2037–2047 (2013).
Sherman, M. H. et al. Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell 159, 80–93 (2014).
Takeda, Y., Tsujino, K., Kijima, T. & Kumanogoh, A. Efficacy and safety of pirfenidone for idiopathic pulmonary fibrosis. Patient Prefer. Adherence 8, 361–370 (2014).
Wang, X. M., Yu, D. M., McCaughan, G. W. & Gorrell, M. D. Fibroblast activation protein increases apoptosis, cell adhesion, and migration by the LX-2 human stellate cell line. Hepatology 42, 935–945 (2005).
Martinez, F. O. & Gordon, S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 6, 13 (2014).
Kim, H. Y. et al. Localized smooth muscle differentiation is essential for epithelial bifurcation during branching morphogenesis of the mammalian lung. Dev. Cell 34, 719–726 (2015).
Shyer, A. E., Huycke, T. R., Lee, C., Mahadevan, L. & Tabin, C. J. Bending gradients: how the intestinal stem cell gets its home. Cell 161, 569–580 (2015).
Selman, M. & Pardo, A. Idiopathic pulmonary fibrosis: an epithelial/fibroblastic cross-talk disorder. Respir. Res. 3, 3 (2002).
Kalluri, R. & Weinberg, R. A. The basics of epithelial-mesenchymal transition. J. Clin. Invest. 119, 1420–1428 (2009). A review of EMT in development and pathology.
Scheel, C. & Weinberg, R. A. Cancer stem cells and epithelial-mesenchymal transition: concepts and molecular links. Semin. Cancer Biol. 22, 396–403 (2012).
Jolly, M. K. et al. Towards elucidating the connection between epithelial-mesenchymal transitions and stemness. J. R. Soc. Interface 11, 20140962 (2014).
Chang, H. Y. et al. Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc. Natl Acad. Sci. USA 99, 12877–12882 (2002).
Hematti, P. Mesenchymal stromal cells and fibroblasts: a case of mistaken identity? Cytotherapy 14, 516–521 (2012).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The international Society for Cellular Therapy postition statement. Cytotherapy 8, 315–317 (2006).
Albrengues, J. et al. LIF mediates proinvasive activation of stromal fibroblasts in cancer. Cell Rep. 7, 1664–1678 (2014).
Avgustinova, A. et al. Tumour cell-derived Wnt7a recruits and activates fibroblasts to promote tumour aggressiveness. Nat. Commun. 7, 10305 (2016).
Rajaram, M., Li, J., Egeblad, M. & Powers, R. S. System-wide analysis reveals a complex network of tumor-fibroblast interactions involved in tumorigenicity. PLoS Genet. 9, e1003789 (2013).
Acknowledgements
The Kalluri laboratory is supported by Cancer Prevention and Research Institute of Texas and the Metastasis Research Center at the University of Texas MD Anderson Cancer Center, Houston, USA. The author wishes to thank V. S. LeBleu for her help in preparing this manuscript and also for help in generating the schematic figures. He apologizes if some citations are missing owing to space constraints.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
MD Anderson Cancer Center and R.K. hold patents in the area of exosome biology and have licensed them to Codiak Biosciences, Inc. MD Anderson Cancer Center and R.K. are stock equity holders in Codiak Biosciences and R.K. receives research support from Codiak Biosciences, Inc.
Glossary
- Desmoplastic reaction, tumour stroma or tumour microenvironment
-
All components of the tumour other than cancer cells. These are generally the components of the host response towards cancer cells, including the immune response. The terms are used interchangeably.
- Basement membrane
-
Organized and assembled extracellular matrix that interacts with all epithelial cells and is also associated with all blood vessels and capillaries.
- Extracellular matrix
-
(ECM). A collection of proteins that are present outside and between cells. ECM consists of large networks of glycoproteins that specialize in protein–protein interactions and serve as ligands of cell surface proteins such as integrins. ECM proteins can form insoluble bundles and generate networks using proteins such as collagens, laminins, entactins, fibronectins and many proteoglycans, such as perlecans.
- Angiogenesis
-
Formation of new capillaries and blood vessels from pre-existing capillaries.
- Cancer-associated fibroblasts
-
(CAFs). All fibroblasts associated with tumours.
- Mesenchymal stem cells
-
(MSCs). Multi-potent stromal cells with the ability to generate connective tissue lineages.
- Wound healing response
-
Injury to the functional parenchyma leads to a reversible host repair or regeneration response that involves myofibroblasts, among other components.
- Quiescent, resting fibroblasts
-
Fibroblast-like cells that are in waiting to become activated when the need arises. They can be considered as resting fibroblasts.
- Fibroblast activation
-
The cellular, biochemical and molecular programme that leads to the conversion of a resting fibroblast into an active fibroblast with a myriad of biological functions.
- Scars
-
The pathologist's term for fibrosis. Fibrosis can be visualized by different dyes that histologically stain different tissue structures, including ECM band-like patterns, making them look like a scar.
- Post-mortem
-
The status of a biological body or tissue after the death of an organism. Observations made in a body or tissue after death are considered post-mortem analyses.
- Fibrosis-associated fibroblasts
-
(FAFs). Activated fibroblasts associated with chronic tissue fibrosis.
- Interstitial space
-
This is the space between layers of functional parenchyma of an organ. It is generally where the support system for an organ exists.
- Tissue fibrosis
-
Chronic wound healing response due to unabated injury to the functional parenchyma.
- Mesoderm
-
The third germ layer, which is considered the precursor to all future mesenchymal cells in the mammalian body, including haematopoietic and connective tissues.
- Resting mesenchymal cell
-
Similar to a resting fibroblast, this is a fibroblast-like cell that is ready to be activated when the need arises. This can also be referred to as an adult tissue-resident mesenchymal stem cell (rMSC).
- Induced pluripotent stem cells
-
(iPSCs). The use of differentiated cells such as fibroblasts to induce stem cell-like features by inserting new genes and/or subjecting cells to physical and biochemical pressures to induce a plastic phenotype.
- Carcinoma in situ
-
(CIS). A basement membrane-contained cancerous lesion. This type of lesion is the lesion that forms early in carcinogenesis. A basement membrane, presumably deposited by CAFs, separates such lesions from the outside interstitial environment.
- Stellate cells
-
Particular mesenchymal cells that are characterized by their vitamin A stores and are found in the liver and pancreas, among other organs. Upon stimulation, they can become activated fibroblasts or myofibroblasts.
- Normal activated fibroblasts
-
(NAFs). Fibroblasts cultured from normal organs.
- Epithelial to mesenchymal transition
-
(EMT). Acquisition of mesenchymal expression programme by epithelial cells.
- Exosomes
-
Extracellular vesicles, approximately 40–150 nm in size, released by all cell types and of multivesicular endosomal origin. They carry proteins and nucleic acids.
- Cancer stem cell
-
Conceptually, a cancer cell that is able to give rise to malignant cancer cells indefinitely and generate tumours; operationally, cancer stem cells are a subset of cancer cells that can, in mice, initiate tumour formation in limiting dilution assay.
- Warburg effect
-
The metabolic phenotype of cells operating predominantly on glycolysis and lactate production despite the bioavailability of oxygen to run oxidative phosphorylation. This is a feature of rapidly proliferating cells, including cancer cells.
- Reciprocal metabolic symbiosis
-
Whereby cancer-associated fibroblasts provide metabolic support to cancer cells in tumours, and vice versa.
- Immunosuppressive TME
-
A tumour microenvironment (TME) that contains cells and other components that interfere with tumour immunity and surveillance.
- Dendritic cells
-
(DCs). Also known as accessory cells, they are antigen-presenting cells in the adaptive immune response.
- T cell anergy
-
Following antigen presentation, T cells may become functionally inactivated, in a hyporesponsive state, to induce immune tolerance.
- T helper 17 cell
-
(TH17 cell). Interleukin-17 (IL-17)-producing T helper cells.
- Migration
-
Movement of cells dependent on motility-inducing molecular signals.
- Regulatory T cell
-
(Treg cell). Regulatory T cells or suppressor T cells maintain immune tolerance to self-antigens and prevent unrestricted effector T cell expansion.
- Drug resistance
-
The adaptive or evasive programmes launched by tumours after treatment with a drug.
Rights and permissions
About this article
Cite this article
Kalluri, R. The biology and function of fibroblasts in cancer. Nat Rev Cancer 16, 582–598 (2016). https://doi.org/10.1038/nrc.2016.73
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2016.73
This article is cited by
-
Deciphering the role of neddylation in tumor microenvironment modulation: common outcome of multiple signaling pathways
Biomarker Research (2024)
-
Integrating single-cell and bulk RNA sequencing data unveils antigen presentation and process-related CAFS and establishes a predictive signature in prostate cancer
Journal of Translational Medicine (2024)
-
A promising target for breast cancer: B7-H3
BMC Cancer (2024)
-
Transcriptomic insights into UTUC: role of inflammatory fibrosis and potential for personalized treatment
Journal of Translational Medicine (2024)
-
CILP is a potential pan-cancer marker: combined silico study and in vitro analyses
Cancer Gene Therapy (2024)