Abstract
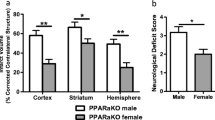
Males and females respond differently to stroke. Moreover, females often experience worse long-term stroke outcomes. Fenofibrate, a peroxisome proliferator-activated receptor alpha (PPARα) agonist has been shown to improve stroke outcome and resolve neuroinflammation in male mice. The present study compares the effect of pretreatment with fenofibrate versus vehicle control in male and female mice during experimental stroke. Mice were treated with low-dose fenofibrate 30 min before and once a day for three additional days after stroke onset. We observed a reduction in infarct volume in male mice 96 h post-stroke with low-dose fenofibrate pretreatment that was due to increase of an M2 macrophage phenotype in the brain and an increase in regulatory cells in the periphery. These outcomes were not replicated in females, likely due to the lower PPARα expression in cells and tissues in females vs males. We conclude that PPARα agonist treatment prior to stroke is neuroprotective in males but not females. These findings indicate PPARα as a probable mechanism of sex difference in stroke outcome and support the need for representation of females in stroke therapy research.





Similar content being viewed by others
Abbreviations
- PPARα:
-
Peroxisome proliferator-activated receptor alpha
- MCAO:
-
middle cerebral artery occlusion
- Treg:
-
regulatory T cell
References
Banerjee A, Wang J, Bodhankar S, Vandenbark AA, Murphy SJ, Offner H (2013) Phenotypic changes in immune cell subsets reflect increased infarct volume in male vs. female mice. Transl Stroke Res 4:554–563. doi:10.1007/s12975-013-0268-z
Bishop-Bailey D (2000) Peroxisome proliferator-activated receptors in the cardiovascular system. Br J Pharmacol 129:823–834. doi:10.1038/sj.bjp.0703149
Bodhankar S, Chen Y, Vandenbark AA, Murphy SJ, Offner H (2013) PD-L1 enhances CNS inflammation and infarct volume following experimental stroke in mice in opposition to PD-1. J Neuroinflammation 10:111. doi:10.1186/1742-2094-10-111
Bodhankar S, Chen Y, Vandenbark AA, Murphy SJ, Offner H (2014) Treatment of experimental stroke with IL-10-producing B-cells reduces infarct size and peripheral and CNS inflammation in wild-type B-cell-sufficient mice. Metab Brain Dis 29:59–73. doi:10.1007/s11011-013-9474-3
Braissant O, Foufelle F, Scotto C, Dauca M, Wahli W (1996) Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, −beta, and -gamma in the adult rat. Endocrinology 137:354–366. doi:10.1210/endo.137.1.8536636
Chinetti G et al. (1998) Activation of proliferator-activated receptors alpha and gamma induces apoptosis of human monocyte-derived macrophages. J Biol Chem 273:25573–25580
Collino M et al. (2006) Oxidative stress and inflammatory response evoked by transient cerebral ischemia/reperfusion: effects of the PPAR-alpha agonist WY14643. Free Radic Biol Med 41:579–589. doi:10.1016/j.freeradbiomed.2006.04.030
Conway SE, Roy-O’Reilly M, Friedler B, Staff I, Fortunato G, McCullough LD (2015) Sex differences and the role of IL-10 in ischemic stroke recovery. Biol Sex Differ 6:17. doi:10.1186/s13293-015-0035-9
Cullingford TE, Bhakoo K, Peuchen S, Dolphin CT, Patel R, Clark JB (1998) Distribution of mRNAs encoding the peroxisome proliferator-activated receptor alpha, beta, and gamma and the retinoid X receptor alpha, beta, and gamma in rat central nervous system. J Neurochem 70:1366–1375
Delerive P, Fruchart JC, Staels B (2001) Peroxisome proliferator-activated receptors in inflammation control. J Endocrinol 169:453–459
Deplanque D et al. (2003) Peroxisome proliferator-activated receptor-alpha activation as a mechanism of preventive neuroprotection induced by chronic fenofibrate treatment. J Neurosci: Off J Soc Neurosci 23:6264–6271
Di Carlo A et al. (2003) Sex differences in the clinical presentation, resource use, and 3-month outcome of acute stroke in Europe: data from a multicenter multinational hospital-based registry. Stroke 34:1114–1119. doi:10.1161/01.str.0000068410.07397.d7
Dotson AL, Wang J, Saugstad J, Murphy SJ, Offner H (2015) Splenectomy reduces infarct volume and neuroinflammation in male but not female mice in experimental stroke. J Neuroimmunol 278:289–298. doi:10.1016/j.jneuroim.2014.11.020
Dunn SE et al. (2007) Peroxisome proliferator-activated receptor (PPAR)alpha expression in T cells mediates gender differences in development of T cell-mediated autoimmunity. J Exp med 204:321–330. doi:10.1084/jem.20061839
Guo Q, Wang G, Namura S (2010) Fenofibrate improves cerebral blood flow after middle cerebral artery occlusion in mice. J Cereb Blood Flow Metab: Off J Int Soc Cereb Blood Flow Metab 30:70–78. doi:10.1038/jcbfm.2009.185
Hurn PD, Subramanian S, Parker SM, Afentoulis ME, Kaler LJ, Vandenbark AA, Offner H (2007) T- and B-cell-deficient mice with experimental stroke have reduced lesion size and inflammation. J Cereb blood Flow Metab: Off J Int Soc Cereb Blood Flow Metab 27:1798–1805. doi:10.1038/sj.jcbfm.9600482
Jalouli M et al. (2003) Sex difference in hepatic peroxisome proliferator-activated receptor alpha expression: influence of pituitary and gonadal hormones. Endocrinology 144:101–109. doi:10.1210/en.2002-220630
Jones DC, Ding X, Daynes RA (2002) Nuclear receptor peroxisome proliferator-activated receptor alpha (PPARalpha) is expressed in resting murine lymphocytes. PPARalpha T B Lymphocytes Both Transactivation Transrepression Competent J Biol Chem 277:6838–6845. doi:10.1074/jbc.M106908200
Kainu T, Wikstrom AC, Gustafsson JA, Pelto-Huikko M (1994) Localization of the peroxisome proliferator-activated receptor in the brain. Neuroreport 5:2481–2485
Lee SR, Kim HY, Rogowska J, Zhao BQ, Bhide P, Parent JM, Lo EH (2006) Involvement of matrix metalloproteinase in neuroblast cell migration from the subventricular zone after stroke. J Neurosc: Off J Soc Neurosci 26:3491–3495. doi:10.1523/jneurosci.4085-05.2006
Li P et al. (2013) Adoptive regulatory T-cell therapy protects against cerebral ischemia. Ann Neurol 74:458–471. doi:10.1002/ana.23815
Liesz A et al. (2009) Regulatory T cells are key cerebroprotective immunomodulators in acute experimental stroke. Nat Med 15:192–199. doi:10.1038/nm.1927
Losey P, Ladds E, Laprais M, Geuvel B, Burns L, Bordet R, Anthony DC (2015) The role of PPAR activation during the systemic response to brain injury. J Neuroinflammation 12:99. doi:10.1186/s12974-015-0295-7
Marx N, Sukhova GK, Collins T, Libby P, Plutzky J (1999) PPARalpha activators inhibit cytokine-induced vascular cell adhesion molecule-1 expression in human endothelial cells. Circulation 99:3125–3131
Moreno S, Farioli-Vecchioli S, Ceru MP (2004) Immunolocalization of peroxisome proliferator-activated receptors and retinoid X receptors in the adult rat CNS. Neuroscience 123:131–145
Mozaffarian D et al. (2015) Heart disease and stroke statistics–2015 update: a report from the American heart association. Circulation 131:e29–322. doi:10.1161/cir.0000000000000152
Offner H, Subramanian S, Parker SM, Afentoulis ME, Vandenbark AA, Hurn PD (2006a) Experimental stroke induces massive, rapid activation of the peripheral immune system. J Cerebr blood Flow Metab: Off J Int Soc Cereb Blood Flow Metabol 26:654–665. doi:10.1038/sj.jcbfm.9600217
Offner H et al. (2006b) Splenic atrophy in experimental stroke is accompanied by increased regulatory T cells and circulating macrophages. J Immunol 176:6523–6531
Ouk T et al. (2013) PPARs: a potential target for a disease-modifying strategy in stroke. Curr Drug Targets 14:752–767
Ouk T et al. (2014a) Effects of the PPAR-alpha agonist fenofibrate on acute and short-term consequences of brain ischemia. J Cereb Blood Flow Metabol: Off J Int Soc of Cereb Blood Flow Metabol 34:542–551. doi:10.1038/jcbfm.2013.233
Planas AM, Chamorro A (2009) Regulatory T cells protect the brain after stroke. Nat Med 15:138–139. doi:10.1038/nm0209-138
Poynter ME, Daynes RA (1998) Peroxisome proliferator-activated receptor alpha activation modulates cellular redox status, represses nuclear factor-kappaB signaling, and reduces inflammatory cytokine production in aging. J Biol Chem 273:32833–32841
Schabitz WR (2013) Regulatory T cells in ischemic stroke: helpful or hazardous? Stroke 44:e84. doi:10.1161/STROKEAHA.113.002228
Seifert HA, Hall AA, Chapman CB, Collier LA, Willing AE, Pennypacker KR (2012a) A transient decrease in spleen size following stroke corresponds to splenocyte release into systemic circulation. J NeuroImmune Pharmacol 7:1017–1024. doi:10.1007/s11481-012-9406-8
Seifert HA et al. (2012b) The spleen contributes to stroke induced neurodegeneration through interferon gamma signaling. Metab Brain Dis 27:131–141. doi:10.1007/s11011-012-9283-0
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC (1998) Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 98:2088–2093
Turner RC, Lucke-Wold B, Lucke-Wold N, Elliott AS, Logsdon AF, Rosen CL, Huber JD (2013) Neuroprotection for ischemic stroke: moving past shortcomings and identifying promising directions. Int J Mol Sci 14:1890–1917. doi:10.3390/ijms14011890
Wang YM, Alexander SI (2009) CD8 regulatory T cells: what’s old is now new. Immunol Cell Biol 87:192–193. doi:10.1038/icb.2009.8
Xiong X, Xu L, Wei L, White RE, Ouyang YB, Giffard RG (2015) IL-4 is required for sex differences in vulnerability to focal ischemia in mice. Stroke 46:2271–2276. doi:10.1161/strokeaha.115.008897
Xu X, Li M, Jiang Y (2013) The paradox role of regulatory T cells in ischemic stroke. Sci World J 2013:174373. doi:10.1155/2013/174373
Zhang W et al. (2008) Soluble epoxide hydrolase gene deletion is protective against experimental cerebral ischemia. Stroke 39:2073–2078. doi:10.1161/strokeaha.107.508325
Zhao BQ et al. (2006) Role of matrix metalloproteinases in delayed cortical responses after stroke. Nat Med 12:441–445. doi:10.1038/nm1387
Acknowledgments
The authors wish to thank Gail Kent for assistance with manuscript submission. This work was supported by NIH/NINDS 5R01NS076013 (HO, JAS). This material is based upon work supported in part by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development. The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of Oregon Health & Science University’s Institutional Animal Care and Use Committee.
This article does not contain any studies with human participants performed by any of the authors.
Funding
This work was supported by NIH/NINDS 5R01NS076013 (HO, JAS). This material is based upon work supported in part by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Biomedical Laboratory Research and Development. The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Dotson, A.L., Wang, J., Chen, Y. et al. Sex differences and the role of PPAR alpha in experimental stroke. Metab Brain Dis 31, 539–547 (2016). https://doi.org/10.1007/s11011-015-9766-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-015-9766-x